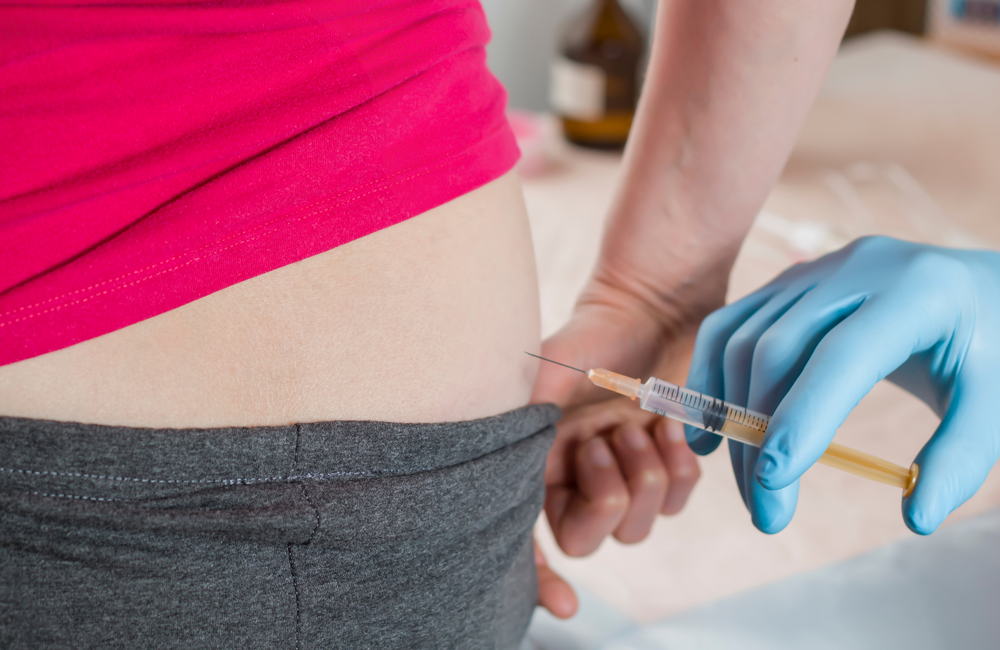
PrEP: history and efficacy
HIV pre-exposure prophylaxis (PrEP), currently taken by about a million people worldwide, is one of the most efficacious ways of preventing HIV infection ever devised.
PrEP involves taking an antiretroviral drug or drugs: antiretrovirals were originally developed to treat HIV infection, but some can be used to prevent people catching it too. In the future, instead of taking a pill, people may be able to have an implant, or wear a patch.
A PrEP injection has already been developed. It is licensed in the US and is expected soon to be available in other parts of the world.
A vaginal ring has also been developed. Though not as effective as oral PrEP in clinical trials, it is licensed for use in Africa. Longer-lasting versions of the ring or ones that combine it with a contraceptive are being developed.
This briefing is mainly about injectable PrEP, though we will look at some other formulations too.
First demonstrated as a concept in animals in the 1990s, studies of PrEP in humans started in 2004. PrEP was initially a controversial idea as people were concerned about side effects and drug resistance. There was also anxiety that, by disincentivising condom use, PrEP might increase rather than decrease vulnerability to HIV.
The first study to prove that PrEP could prevent HIV was in 2010. The iPrEx study found an antiretroviral pill containing the two drugs tenofovir disoproxil and emtricitabine (TDF/FTC), given to gay and bisexual men daily, reduced their risk of HIV infection by 44%, compared to a placebo pill.
Eighteen months later, the Partners PrEP study found that PrEP given to the HIV-negative partner in heterosexual couples reduced their risk of infection by 65-70%. Partners PrEP, and two other early studies, also tested tenofovir on its own, but efficacy was in most cases better if the two drugs were used.
The US Food and Drug Administration decided in 2012 that this efficacy was enough to license the TDF/FTC pill (then known by the brand name of Truvada) for prevention as well as treatment. PrEP use took off slowly but started to expand after the US Centers for Disease Control and Prevention issued recommendations for its use in 2014. An estimated quarter of a million people, mainly gay men, have used it in the US.
In Europe, PrEP was not licensed until the more conclusive results of the PROUD and IPERGAY studies appeared in 2015. Both studies – the latter using ad-hoc, event-driven TDF/FTC PrEP rather than daily – showed that PrEP reduced the risk of HIV infection in trial subjects by 86%. The European Medicines Agency recommended PrEP for approval in 2016.
The World Health Organization has supported PrEP since 2012. In recent years, usage has greatly expanded worldwide, especially in sub-Saharan Africa, which now accounts for the majority of PrEP users.
Does PrEP prevent the spread of HIV?
Oral PrEP is clearly efficacious on an individual level. For a while it was unclear whether it was effective on a population level: whether it was contributing to overall reductions in the rate of HIV infections.
Part of the reason population-level efficacy is hard to prove is because of the widespread endorsement in 2015 of universal and immediate treatment of people with HIV upon diagnosis. ‘Treatment as prevention’, based on incontrovertible evidence that people with an undetectable viral load could not pass on HIV, found itself enshrined in the 90-90-90 target and is the subject of the U=U campaign.
Significant falls in HIV incidence, especially among gay and bisexual men, in countries such as the UK and France were initially attributed to there being fewer people with HIV with a transmissible viral load.
To demonstrate clearly that PrEP pills were contributing to the fall in infections, there needed to be a country or population in which 90-90-90 targets were achieved quite early, and largely before PrEP was introduced. If in that country a two-phase reduction in HIV incidence could be seen – the first accompanying universal treatment and then a steeper downward trend following the adoption of PrEP – this would provide evidence that PrEP had its own separate effect on a population level, and could make it possible to quantify that effect.
The first such evidence came from New South Wales in Australia, where the launch of a statewide PrEP campaign was followed by a one-third decline in HIV diagnoses in the state’s gay population.
A subsequent study came from Scotland, which in July 2017 was the first nation in the UK to provide PrEP via the NHS. Scotland adopted universal treatment as prevention in 2015 and falls in HIV diagnoses due to viral suppression among people with HIV had already been seen. But further falls seen in the two years after the introduction of PrEP – of 20% in the gay population as a whole, and 43% in gay men attending sexual health clinics – were attributed to PrEP.
A study from the US found that HIV diagnoses in gay men had fallen in cities with the highest proportion on PrEP, independently of the effects of treatment as prevention. And a 2022 French study which compared rates of HIV diagnosis in men who had, and had not, used PrEP found a population-level effectiveness of 60%, rising to 93% in the most consistent users.
It is difficult to gauge a separate, population-level effect of PrEP in countries where falls in incidence have yet to occur or where expansions of PrEP and treatment are happening at the same time. But PrEP may lead to distinct falls in incidence in specific populations. One example is a west African study comparing HIV incidence in men who took PrEP and men who did not, which found substantially lower rates in PrEP users.
Several subsequent studies have found that HIV incidence in gay men started falling before the introduction of PrEP – in both Australia and the UK. The truth is probably that in places where antiretrovirals are accessible both for treatment and for prevention, they both play a role in reducing the transmission of HIV, and maybe a synergistic one, as this UK-based modelling study suggests.
Why doesn’t PrEP always work?
The biological efficacy of oral PrEP – its ability to prevent HIV infection if taken completely consistently and appropriately – is probably close to 100%. There have been fewer than 20 cases worldwide of HIV infections occurring in people with documented good adherence to PrEP, and no more than ten in people taking oral PrEP.
As the pivotal studies made clear though, PrEP’s efficacy is highly dependent on whether people take it when needed. PrEP is fairly ‘forgiving’ in that, for anal sex at least, it is estimated that the equivalent of four or more doses of PrEP per week (57% adherence) is enough to confer sufficient levels of protection.
This implies reasonable consistency of use, though. HIV infection is much more likely if PrEP is stopped for weeks or months, and if these PrEP breaks do not coincide with gaps in ‘seasons of risk’. In the iPrEx study, it was estimated that efficacy would have been up to 95% if trial participants had taken it consistently.
Low adherence was the cause of PrEP failure in several studies, especially among women. In the Fem-PrEP study among young women in Africa, PrEP had zero efficacy. Adherence as measured by pill count was 85%, but as measured by drug level was only 28-40%. In the VOICE study, self-reported adherence was 90% but adherence measured by drug levels was only 29%.
A qualitative analysis of the VOICE study found that participants had varied reasons not to take PrEP pills. Some women joined the trial because they wanted its ancillary benefits such as STI and pregnancy care, rather than PrEP. Many were conscious they were in a placebo-controlled trial and decided to only start taking PrEP if its efficacy was proven. Others had reasons unrelated to being in a trial, such as fear of side effects, unstable housing or lack of privacy, or concern that partners or friends might think they had HIV if they saw pills. Another common reason was that participants did not believe themselves to be at high enough risk to need PrEP, even in situations where they clearly were, because HIV infection was something ‘other’ women got.
Women are not the only people who sometimes have difficulty with adherence to PrEP, and low adherence seems tied to relative social inequality. The ADAPT study, which compared daily PrEP to event-based dosing, looked at PrEP adherence in largely employed and educated gay men in Bangkok, largely unemployed and poor Black gay men in New York, and largely unemployed and poor women in Cape Town. The South African women had poorer adherence to PrEP than the Thai men – but the New York men had the poorest adherence of all, especially to event-driven regimens. A lot of the poor adherence seemed to be driven by not having the post-sex doses to hand.
But it is more often women who seem to find that oral PrEP is not the prevention answer that suits them. PrEP projects targeting female sex workers and adolescent girls and young women have often had poor recruitment, poor retention and poor adherence.
Drawing all this evidence together, a global meta-analysis of PrEP adherence rates found that 41% of people who started taking PrEP stopped taking it within six months (though just under half re-started it at a later point). The six-month discontinuation rate was even higher in cisgender women and heterosexual men at 72%.
The aspiration that lies behind the development of long-acting forms of PrEP is that not having to think about taking PrEP more than once every few months would improve adherence rates enough for it to reach community-level effectiveness in all populations.
Drug levels: getting PrEP to the parts of the body that need it
Poorer adherence is not the only reason PrEP may work less well in some people. There may be biological reasons too.
Levels of the two drugs usually used for oral PrEP – tenofovir disoproxil (TDF) and emtricitabine (FTC) – take longer to reach saturation level (‘steady state’) in the epithelial cells lining the vagina and cervix than in the rectum. This implies that oral PrEP may provide better protection of anal sex than of vaginal sex.
Early studies of drug levels in cervical and vaginal tissue found levels of tenofovir diphosphate (TFV-DP – the intracellular, metabolised version of the ‘prodrug’ TDF) that were only a tenth to a hundredth as high as levels in rectal tissue. A 2016 modelling study found that a minimum adherence to six out of seven doses of TDF/FTC a week (85%) was needed to protect female genital tract tissue from HIV, while adherence to as few as two doses a week (28%) was sufficient to protect colorectal tissue. A third study found that it took 10 to 12 days for TFV-DP to approach maximum levels in cervical epithelial cells, but only five days in rectal cells.
Two other studies showed that infections in gay men who took the equivalent of four doses of TDF/FTC a week were rare, but were more common in women, and that six to seven doses a week were needed to reach the same level of protection. These drug level studies have served ever since as the basis for PrEP guidelines that rule out event-based, ‘2-1-1’ dosing for vaginal sex.
The newer prodrug formulation of tenofovir, tenofovir alafenamide (TAF), was developed by Gilead Sciences as a way of reducing side effects due to high levels of tenofovir in the blood. It is metabolised faster and so reaches higher levels in cells more quickly than TDF, while blood levels are lower. In two paired studies, levels of TFV-DP in lymphocytes (cells) were about seven times higher after taking TAF than after taking TDF.
This implies that TAF may be more effective in women, but randomised trial data comparing TAF-based PrEP and TDF-based PrEP in women will not be available until 2024. The DISCOVER trial comparing the two formulations in gay men and transgender women found that there was a trend towards greater efficacy in participants taking TAF, but this was not statistically significant.
Regarding transgender people, there have been concerns that high levels of gender-affirming hormones might affect PrEP drug levels, and vice versa, in both women and men. Several studies showed that feminising hormone levels in transgender women were very little affected by PrEP, but that some, though not all, studies showed that use of feminising hormones was associated with slightly lower tenofovir levels. One of the most carefully designed studies (iBREATHE) found levels of TFV-DP that were 30% lower in transgender women than in cisgender women. This is higher than the four-doses-a-week level thought to be efficacious in gay and bisexual men, but not quite as high as the six-doses-a-week level thought to be needed for cisgender women. Levels in trans and cis men did not differ from each other.
For now, although it is plausible that the efficacy of oral PrEP in trans women practising receptive anal sex may be sufficient to allow event-based dosing, it is wise to err on the side of caution and take PrEP daily.
Long-acting drugs for other medical conditions
Long-acting drug formulations have a long history. They include:
- extremely long-acting pills
- ‘depot’ formulations, which involve injecting the drug into a muscle or fatty part of the body, where it is slowly and evenly absorbed
- subcutaneous injections – smaller ones injected just under the skin
- skin patches or transdermal implants (the latter use tiny projections that pierce the skin but are too small to be felt)
- implants that are inserted under the skin
- vaginal rings.
An advantage of patches, implants or rings is that they can be removed if they cause side effects or if, as might be the case with contraceptives, their effect is no longer desired.
A depot formulation of penicillin was introduced as long ago as 1949. Injectable antipsychotic drugs were first introduced in the 1960s for patients who for obvious reasons might find adherence to regular oral medications difficult.
Injectable forms of the masculinising hormone testosterone were developed in the 1950s; transdermal patches started to be used in the early 2000s. The hormone agonist leuprorelin – an anti-androgen used to suppress prostate cancer, as a component of gender-affirming therapy, and as a puberty blocker – was first introduced as a depot formulation in 1989; there is now one lasting six months.
The drugs most comparable with injectable PrEP however are the injectable contraceptives. The first one to be introduced was Depo-Provera, a depot formulation of progesterone, in 1960. Used alone, progestin-based contraceptives can now be given every two to three months. As of 2019, injectable contraceptives were used by 74 million women worldwide and were the most popular method in sub-Saharan Africa.
Having a range of contraceptive methods available increases the likelihood that users will find a method that corresponds to their needs and preferences. Some formulations of contraceptives last even longer than the injectables and similar products are being studied as ways of giving PrEP.
The contraceptive implant is a small rod of plastic containing a progesterone analogue that is inserted under the skin and lasts for up to three years. It has been available in the US since 2006 and by 2010 was already approved by more than 90 countries. It is currently used by 23 million women worldwide.
The vaginal contraceptive ring, which contains progesterone and oestrogen analogues, was approved in the US and Europe in 2001. It fits over the cervix and is designed to be used for three weeks out of the four of the menstrual cycle. Research is now underway into a combination ring to provide both contraception and PrEP.
The US Office of Women’s Health has calculated the average efficacy of different methods of birth control. The chance of getting pregnant within a year for a woman trying for a baby is about 86% before the age of 35. Using condoms reduces this chance to 18% (79% efficacy), the oral pill and the vaginal ring (in typical use) to 9% (91% efficacy), contraceptive injections to 6% (93% efficacy), and the contraceptive implant to 1% (99% efficacy).
The development of long-acting HIV treatment
Although there has long been interest in developing longer-acting antiretrovirals, it took some time to manufacture the first long-lasting injectable formulation. Most antiretrovirals required doses too large – in the order of 200-800mg a day – to make depot formulations feasible.
However, the effective daily dose of the non-nucleoside reverse transcriptase inhibitor rilpivirine was only 25mg a day, which allowed an injectable formulation, with the drug absorbed into virus-size nanoparticles, to start human trials in 2007.
The second drug to be successfully developed as a long-acting injectable formulation was cabotegravir, which is the one now approved for PrEP. Cabotegravir is a longer-lasting relative of the integrase inhibitor dolutegravir, which itself only required a 50mg daily dose. In 2012, cabotegravir’s strong binding to proteins suggested that it could be given as an injection every month or even every two months.
Starting in 2014, the LATTE trial tested rilpivirine and cabotegravir together, initially as tablets, and then as injections given every four weeks. In 2016 it showed that the rate of viral suppression was the same for oral and injectable formulations. LATTE was followed, in 2019, by the larger ATLAS study in treatment-experienced patients and FLAIR study in people new to treatment, both comparing the injectables to oral HIV treatment. The proportion of people maintaining an undetectable viral load (about 94%) was statistically the same in both studies, whether they were on injections or pills.
These results were good enough for the European Medicines Agency to grant regulatory approval of injectable cabotegravir and rilpivirine (sold under the separate brand names Vocabria and Rekambys) in October 2020. The US Food and Drug Administration (FDA) gave its approval in January 2021 to the two injections, which are sold together as Cabenuva in North America.
Regulatory approval does not guarantee availability however – it just declares that the drug is safe and effective. Some countries also ask health technology agencies such as the UK’s National Institute for Health and Clinical Excellence (NICE) to evaluate the drug’s cost-effectiveness. The approval in November 2021 by NICE of Vocabria and Rekambys was a step nearer to seeing it available in the clinics in 2022.
A comparable instance in PrEP was that though the US FDA approved it as safe and effective to use in July 2012, it was not till the US Centers for Disease Control and Prevention issued detailed recommendations for its provision two years later that PrEP started being used by more than a handful of people. Similarly, it will take more than regulatory approval for injectable PrEP to be widely used.
The development of injectable PrEP
Soon after injectable rilpivirine was developed for treatment, studies started of its potential use for prevention, but eventually concluded that rilpivirine would not work on its own for PrEP as its half-life was not long enough.
However, the lengthy half-life of cabotegravir in tissues suggested it could also be used in PrEP. It also suggested that the levels it reached should be sufficient to work as a prevention drug when used alone.
Studies of cabotegravir, then known as GSK744, started in monkeys in 2013 and in humans in 2015. By 2017, the HPTN 077 study had taken place in 199 people at low risk of HIV infection. Drug level measurements indicated that one injection every eight weeks was likely to be sufficient to ward off HIV infection.
Because of these positive results, two large efficacy studies were launched: HPTN 083 in gay and bisexual men and transgender women in the US, Latin America, South Africa and south-east Asia, and HPTN 084 in cisgender women in seven countries in sub-Saharan Africa.
The results of these two studies were far better than anyone expected. Whereas ATLAS and FLAIR had shown that injectable antiretroviral therapy was roughly equivalent to oral antiretroviral therapy in terms of its ability to suppress HIV, injectable cabotegravir was several times more effective than oral PrEP in its ability to prevent infection.
In HPTN 083, whose results were announced in February 2020, among 4600 gay and bisexual men and transgender women, there were 38 infections among people taking tenofovir disoproxil/emtricitabine (TDF/FTC) pills, but only 12 in people receiving the injections. This equated to a 69% reduction in the likelihood of an HIV infection, over and above the preventative efficacy of oral PrEP. Final figures revised this to 41 infections in people taking oral PrEP and 14 in people receiving cabotegravir – an efficacy of 66% over TDF/FTC oral PrEP.
Efficacy was almost exactly the same in cisgender men and transgender women at 65% in cis men and 66% in trans women, though the latter result was not statistically significant because of the smaller number of trans women in the study.
In HPTN 084, the results announced in November 2020 were even more conclusive. Among 3223 cisgender women, there were 34 HIV infections in women allocated to take TDF/FTC pills, but only four in women receiving cabotegravir injections – an 89% reduction in the likelihood of infection, over and above any effect of oral PrEP.
On the basis of these studies, the US FDA granted approval of cabotegravir, sold under the brand name Apretude, to be used as PrEP in the US. So far this is the only regulatory agency to approve cabotegravir as PrEP.
Soon afterwards, revised figures from HPTN 084 established that there had been 36 infections on TDF/FTC rather than 34, and that one of the four infections in the participants allocated to cabotegravir had in fact happened before she joined the study. This raised the efficacy of cabotegravir in preventing HIV to 92% better than TDF/FTC.
Breakthrough infections
One potential problem of injectable PrEP is what happens when you stop receiving injections. The levels of injectable drugs last a long time in the body – in some cases, especially in women, over a year. This creates the conditions whereby, if someone is exposed to HIV and the virus is able to overcome the waning drug levels and infect the person, the viral population that is ‘selected’ – which is able to produce more virus – is the one with resistance mutations.
This did happen in the 2012 study of rilpivirine, where a participant caught HIV that was either already drug resistant or became drug resistant after a single injection of the drug and a single sexual exposure to HIV 40 days later. The participant tested HIV positive three months after her PrEP injection, but retrospective testing of blood samples found she had a detectable viral load two months after her PrEP injection, and just two weeks after infection.
In the randomised controlled studies, while the superior effectiveness of injected cabotegravir as PrEP was obvious, there were some infections in people who received the injections.
In HPTN 083, including an open-label extension in which all participants remaining in the study were offered the injections, there were 25 infections in people having injected PrEP and 72 in people taking oral PrEP.
Out of the 25 infections in people allocated to cabotegravir, 12 were in people who had left long gaps between doses. Three had missed their injection appointments by shorter periods of time – between one and 3.6 months. Three were in people who caught HIV during the five weeks they were taking cabotegravir pills as a ‘lead in’ before beginning the injections.
This left seven breakthrough cases who were infected despite attending regularly for their injections and appearing to have consistently high drug levels. At the moment we have no explanation for this other than that cabotegravir may be biologically slightly less efficacious, or develop lower-than-average levels, in a small minority of people.
The HIV in all seven people with breakthrough infections developed drug resistance to cabotegravir. A contributing factor was probably that, as has been seen with other breakthrough infections on both oral and injected PrEP, the drugs suppressed the normal HIV viral load to such an extent that HIV antibodies took a very long time to appear.
Tests for HIV antibodies repeatedly returned false negative results. Retrospective RNA (viral load) testing of blood samples showed that if RNA tests had been used routinely, their infections could have been detected seven to 20 weeks earlier, which might had stopped them developing drug resistance. However RNA tests would add to the already greater expense of injectables.
It is notable that no breakthrough infections and no cases of cabotegravir resistance have been seen in the women in HPTN 084.
The vaginal ring
While attention often focuses on injectable cabotegravir, the dapivirine vaginal ring is another proven form of long-acting PrEP. However, it is also an example of how tortuous the process towards full provision can be.
It was not long after the approval of the contraceptive ring that, in 2004, Johnson and Johnson signed over its patent rights on a non-nucleoside reverse transcriptase inhibitor dapivirine (then called TMC120) to a new research consortium, the International Partnership for Microbicides (IPM), to investigate it for use in a vaginal ring.
After a long process of fundraising and early studies, RING and ASPIRE, two phase III studies of the HIV PrEP vaginal ring announced their results in February 2016. Both reported rather disappointing efficacy compared with oral tenofovir disoproxil/emtricitabine (TDF/FTC) PrEP. Efficacy rates in RING and ASPIRE were 31% and 27%, respectively: the ring only prevented a quarter to a third of HIV infections that would otherwise have happened. Efficacy was better in slightly older women – at 56% in women aged 22-26 in ASPIRE. Later data showed that with high adherence, efficacy was as high as 65%. Qualitative studies showed that adherence tended to improve over time.
Two years later, open-label extensions of these studies, renamed DREAM (for the RING study) and HOPE (for the ASPIRE study) reported. They found that the proportion of women with low adherence had fallen. Because these were no longer placebo-controlled studies, it was only possible to measure HIV incidence against a model of what incidence would have been if the rings had not been used. Effectiveness came out as 62% in DREAM but only 39% in HOPE.
The vaginal ring offered women an alternative to oral PrEP, especially as studies showed that efficacy was not the sole factor women took into account when choosing an HIV prevention option. Though approval rates were high in any case, many said they would prefer a product with moderate efficacy that offered ease of use and discretion to a product that was highly efficacious but hard to use or obvious to sexual partners. The vaginal ring was already becoming a political issue, with activists from Africa and elsewhere protesting at conferences in favour of its approval.
So there was qualified relief when in July 2020 the European Medicines Agency (EMA) gave regulatory approval to the ring, though in an unusual way: it was done under a measure called Article 58, which enabled the regulatory expertise of the EMA to be used to grant a licence for a device intended to be used in lower-income countries. The World Health Organization (WHO) followed it immediately with their recommendation that the ring should be offered as an additional prevention choice. But the ring was not licensed in Europe or other high-income countries.
In December 2021, the FDA advised IPM that it would be unlikely to give approval to the ring in the US: this decision appeared to be based on the existing availability of oral PrEP. IPM and WHO declared their disappointment, while advocates for Black women in the US decried the decision as one that would rebound to Africa. Indeed, only a few days after South Africa’s licensing agency granted approval, the country’s Ministry of Health ordered a review of effectiveness. The advocacy organisation AVAC has started a campaign for the ring’s approval and an open-letter statement urging accelerated provision has been composed by a global panel of HIV prevention advocates.
Future directions
Injectable cabotegravir will not be the final word in long-acting PrEP. Even as it was being developed, interesting new long-acting drugs of other types were being developed.
Until recently one of the most promising was islatravir. This drug’s potency is “several orders of magnitude better” than similar nucleoside-based drugs such as tenofovir. This was because it inhibits HIV not in one, but two different ways and because a single oral dose has a half-life of over a week, implying it could be dosed even less often than that.
Islatravir was an obvious candidate for long-acting formulations, with a weekly pill investigated for HIV treatment. Islatravir looked even more promising as a candidate for PrEP. In 2013, monkeys given an injection of islatravir PrEP every three months were completely protected from HIV infection. Human trials started in 2017, followed in 2021 by the IMPOWER studies that evaluated islatravir as a monthly pill that only needed to be taken once a month, in comparison with daily TDF/FTC.
An implant for insertion under the skin that might only need to be replaced once a year was also being investigated. An advantage of an implant – a technology already used in contraceptives – is that, unlike an injection, it can be removed, in the case of unexpected toxicity or patient preference.
Unfortunately, however, it was found that islatravir was associated with falls in CD4 and total lymphocyte counts, and in December 2021 US authorities ordered all studies of islatravir to be put on clinical hold. At the moment it looks unlikely that islatravir research can be continued. Other drugs with a similar mode of action are at the preclinical stage of investigation but it is unknown whether the effect seen is specific to islatravir or would affect other drugs in the same class.
In 2017 the development of lenacapavir, the first capsid inhibitor, was announced. This drug was capable of being administered as a subcutaneous (under the skin) injection every six months, of far less bulk than the cabotegravir injections. It proved to be efficacious in studies both as a first-line HIV treatment or in people with extensive drug resistance in combination with other antiretrovirals.
For PrEP, lenacapavir’s manufacturer Gilead Sciences have started the two PURPOSE studies. PURPOSE 1, being conducted with cisgender women in South Africa and Uganda, will compare twice-yearly lenacapavir injections, daily TDF/FTC, and daily tenofovir alafenamide (TAF)/emtricitabine. As well as testing lenacapavir, the study will also be the first-ever direct comparison of the two tenofovir formulations as PrEP in women. PURPOSE 2, in US gay and bisexual men and trans women, will compare twice-yearly lenacapavir injections against daily TDF/FTC.
Gilead is also developing an injectable formulation of TAF, and a TAF implant has already shown efficacy in monkey studies.
Immunotherapies
A number of companies and university researchers are also looking at the use of immunotherapies for HIV prevention and treatment. While antiretrovirals are chemicals that block the activity of specific viral proteins, immunotherapies are derived from or mimic the body’s own natural antiviral defences.
At present these mainly consist of passively administered broadly neutralising antibodies (bNAbs). Antibodies are a vital component of the immune system, soluble proteins that are made in bulk when the body encounters either a new pathogen or (faster and more efficiently) when it encounters one it has encountered before.
In most infections the antibody response helps to successfully rid the body of the infection – either naturally in the course of an infection, or because a vaccine has already primed the immune system.
However in HIV, although antibodies are produced in response to infection, they are too weak and non-specific to contain the infection, which is one reason why HIV infection is lifelong – and why, so far we have not produced an effective HIV vaccine.
But in 10-30% of people, highly mutated antibodies develop after some years of infection. They are ineffective at this stage but, if given to non-infected people, would be capable of blocking or containing HIV.
These antibodies have two modes of operation: firstly, they directly block (‘neutralise’) viral infection by attaching themselves to different parts of the envelope protein HIV uses to gain entry to cells. Secondly, their presence alerts the other parts of the immune system to spring into action and purge viral particles or infected cells. In theory this could mean that they could form part of a cure for HIV.
Long-acting formulations of antibodies can be made for dosage by infusion (or, in the future, via vaccines that stimulate antibody production). In HIV treatment a series of experiments using a dual combination of bNAbs has achieved incrementally increasing timespans during which recipients have been able to stay off antiretroviral therapy, as in this study from 2018 compared with this one from 2022. Children born with HIV appear to be good candidates for antibody therapy, partly because their immature immune systems and short time infected means that their HIV has not yet developed the ability to escape immunotherapies.
In the case of bNAbs for PrEP, there has so far been one large randomised controlled study, the AMP study. This used a single antibody – not something that will be done again – and only produced a 19% reduction in HIV infections, which was not statistically significant. It was, however, 75% effective in the type of viruses most sensitive to the particular antibody used. As a proof of concept it showed that bNAbs could be used to prevent HIV.
Studies of more potent combinations of antibodies are planned, but AMP underlined the Achilles heel of bNAbs – HIV acquires resistance to them easily and many types of circulating HIV already have pre-existing resistance. Scientists hope that more potent and broad-spectrum combinations of antibodies can be found but they may have to be dosed in combination with other drugs and immunotherapies to reach full potential. This full potential will only come when we have vaccines that work by inducing the body to make its own antibodies against HIV. As well as the Scripps vaccine noted above, there are a number of other vaccines under development, some of which use the same RNA technology as some COVID vaccines.
Practicality and cost-effectiveness
The HPTN 083 and 084 studies have demonstrated convincingly that injectable PrEP could be the most effective HIV prevention method yet devised. However, the fact that at the time of writing an application to license injectable PrEP in Europe has still not been made is a reminder of the many barriers to it achieving its full potential.
One of the attractive features of oral PrEP is that it involves cheap generic drugs that, in theory at least, can be accessed easily or informally, though it is still important that PrEP recipients test for HIV and STIs regularly. During the COVID-19 epidemic, many oral PrEP programmes were maintained and some even increased their reach, with more use of telemedicine, home delivery, home testing and NGO provision.
Compared with this, injected PrEP, at least initially, will require new and expanded clinical services. Firstly, clinics will need to assess people’s need for injected PrEP and triage patients accordingly – particularly in the early stages when it is likely to be considerably costlier and scarcer than oral PrEP. Secondly, they will need to train staff to administer the injections (the intramuscular injection of cabotegravir requires a specialised technique), and find the time for them to do it. And thirdly, they will need admin backup to schedule injection appointments, send reminders, and trigger intervention for people who are late for a dose.
An early adopter in Canada has detailed the extra clinical burden of providing injectable treatment – a burden that his clinic could not cover. In this case, giving the injections was taken on by a detached nursing service operating out of local centres, which was funded by the relevant pharma company. With PrEP the increase in cost and time is likely to be relatively greater as oral PrEP is a lighter-touch service.
Another challenge for any new prevention method is that is needs to be good value for money, in comparison with existing products. In the case of PrEP, the point of comparison is the generic version of oral TDF/FTC, which is widely available for less than $350 a year.
Selling the new drugs at prices that are too high will put them out of reach. A 2021 cost-effectiveness analysis found that an injectable treatment regimen would need to cost no more than $131 a year – about 68% more than the cheapest oral antiretroviral therapy regimen – in order to be cost-effective in Africa. An analysis of PrEP found that even in Europe, injectable cabotegravir PrEP would have to cost no more than US$3000 a year. At the time, the Canadian list price for Cabenuva was $25,800 a year, so the price of injectable PrEP will clearly have to fall a lot further for it to be available in more than exceptional circumstances.
In May 2022 the company that manufactures injectable cabotegravir, ViiV Healthcare, announced that it will license its patents with the UN-backed Medicines Patent Pool. This may help enable at-scale access to injectable PrEP in low- and middle-income countries. But injectable and long-acting PrEP may have to jump further hurdles if it is to reach its enormous potential. As a recent article by three top researchers in South Africa put it, “What is the use of anti-HIV injections when those who need it most can’t use it?”
