This page is about injectables used for HIV treatment, rather than prevention of infection (PrEP). We have another page on injectable cabotegravir for PrEP.
Which medications can be taken by injection?
The first complete HIV treatment provided by long-acting injections is a combination of two medications. Cabotegravir belongs to the same class of medication – integrase inhibitors – as the widely used HIV medication dolutegravir. The second medication is called rilpivirine and is already used for HIV treatment in tablet form. It is from a class of drugs known as non-nucleoside reverse transcriptase inhibitors (NNRTIs).
In Europe, the brand name for injectable cabotegravir is Vocabria, while the brand name for injectable rilpivirine is Rekambys. In North America and Australia, the two drugs are packaged together, with the brand name of Cabenuva.
Cabotegravir and rilpivirine can be taken either once a month or every two months. For more information on this, see How frequent are the injections? on this page.
There are also some other injectable HIV treatments now available in a small number of high-income countries, but only for people who have resistance to other HIV medicines.
The first is lenacapavir (Sunlenca) which is the first HIV capsid inhibitor. Lenacapavir is given by injection once every six months, but it needs to be taken in combination with other HIV medicines (usually daily tablets) to work effectively. There are also studies to see whether lenacapavir can be used by HIV-negative people as injectable PrEP.
The second is ibalizumab (Trogarzo) which is a monoclonal antibody. It is given as a two-weekly injection or infusion, and it also needs to be taken in combination with other HIV medicines to work effectively.
Pharmaceutical companies are researching other long-acting medications. See the last section of this page for more information.
This information page focuses on cabotegravir and rilpivirine as it is currently the most widely available injectable treatment. For more information about cabotegravir and rilpivirine, take a look at our factsheet and our detailed page in the A-Z of antiretroviral medications.
Where is injectable treatment available?
Injectable cabotegravir and rilpivirine have been approved by drug regulators in Argentina, Australia, Botswana, Canada, the European Union, Israel, Japan, Russia, Switzerland, United Kingdom, United States, and United Arab Emirates. In many of these countries however, the injections are not yet available in clinics and decisions on reimbursement still need to be made.
In the United Kingdom, NHS clinics began providing injectable cabotegravir and rilpivirine in 2022. Elsewhere in Europe, countries with access include France, Germany, Switzerland, Sweden, Norway, Finland, Denmark, Belgium and The Netherlands.
They are not yet available in the global south.
Are injections better than tablets?
No, their efficacy is comparable. The cabotegravir and rilpivirine injectable regimen was approved by drug regulators based on the results of three randomised control trials.
- The first study involved people who hadn’t taken HIV treatment before. All started with daily pills for around four and a half months (20 weeks to be precise). Half then continued with daily pills, while half switched to monthly injections.
- The second study recruited people who were already taking HIV treatment and had an undetectable viral load. Half continued with their daily pills, while half switched to monthly injections.
- As a follow-on from the second study, the third study compared injections once a month with injections once every two months.
Each of the studies assessed the success of treatment in terms of how many people had an undetectable viral load after just under a year. Pooling the results of the three studies, this was the case for 94% of people given the injections. The first two studies found that the monthly injections were as effective as daily pills. (In research terminology, they were ‘non-inferior’.) Similarly, the third study found that injections every two months were non-inferior to monthly injections. Long-term follow-up of the second and third studies found that the injectable treatments (both monthly or every two months) remained effective after three years.
Unlike with tablets, these studies identified that there is a very small risk of treatment failure with injectable treatment even when injections are consistently taken on time. The failure rates among those receiving two-monthly injections in the third study were 1 in 70 (1.4%) in the first year, and 1 in 60 (1.6%) in the second year. If your treatment fails, this means your viral load will go up and you may develop resistance to the drug types used in injectable treatment. Although the risk is very low, this may influence your decision about switching to injectable treatment.
Visit the A-Z of antiretroviral medications to find out more about these research studies.
Who can take them?
Injectable cabotegravir and rilpivirine are licensed for adults with a viral load under 50 on a stable oral antiretroviral regimen, and without any evidence of resistance to integrase inhibitors (like cabotegravir) or NNRTIs (like rilpivirine). That could potentially be a large number of people currently taking HIV treatment.
In the US, they are also licensed for use in young people aged 12 or older who weigh at least 35kg.
However, they are not currently licensed for people who have a detectable viral load, which is likely to include those who have difficulties with adherence to daily pills or whose current HIV treatment is not working well.
Also, they cannot be used for someone starting HIV treatment. If someone new to HIV treatment does want to use them, they would need to take an oral antiretroviral regimen for a few months first and achieve viral suppression on that, before switching to the injections.
They are not currently licensed anywhere for children under 12 or during pregnancy or breastfeeding, as the clinical trials on which the approval licences are based did not include these groups. Research into use of injectable cabotegravir and rilpivirine by children and pregnant people is currently ongoing.
They are also not suitable for people who have hepatitis B co-infection (who should take a drug combination that includes medications that are active against both HIV and hepatitis B).
Having a strain of HIV called A6, a body mass index over 30 and buttock implants or fillers all increase the risk of injectable cabotegravir and rilpivirine not working effectively. Your doctor will take these risk factors into account when considering your eligibility for injectable cabotegravir and rilpivirine and decide with you whether the benefits of switching to injectable treatment outweigh the risks.
Are injectable treatments recommended in guidelines?
HIV treatment guidelines issued in several high-income countries recommend injectable cabotegravir and rilpivirine for select groups of people living with HIV. For example, this is the case for guidelines in the United Kingdom, United States and Europe. However, guidelines from the World Health Organization – influential in the global south – do not currently recommend injectable cabotegravir and rilpivirine.
In current guidelines, injections are an option for people who are already taking HIV treatment and have a viral load below 50. People may be able to switch to the injections if there is nothing in their medical history which would increase the risk of the injections not working. Injections are not an option for people starting treatment for the first time. In addition, the UK guidelines prioritise the injections for individuals who have a particular need for them, for example people who have psychological barriers to taking pills every day.
Who might get the most benefit from injectable treatment?
Surveys suggest that large numbers of people living with HIV would be interested in trying injectable treatment. Nonetheless, providing injections requires extra staff and resources, so they won’t necessarily be made available to all people living with HIV, but may be prioritised for individuals who are most likely to benefit.
Many people with HIV are fed up with taking pills every day or find it a constant reminder of their HIV status. For people who wish to keep their HIV status private, switching to injections may increase confidentiality and privacy. For example, they could help people who live in shared accommodation and currently feel the need to hide their medication. There could be advantages for people who are homeless, in prison or who use drugs. Injectable HIV treatment will suit people who are able to stick to clinic appointments, even if they struggle with daily pills. However, cabotegravir and rilpivirine is not yet approved for people with detectable viral loads.
The injections may be practical for people who are travelling, especially to countries which restrict entry to people living with HIV. They may also provide an option for people who have specific challenges such as swallowing difficulties, drug malabsorption or gastrointestinal conditions.
For some other people living with HIV, the disadvantages may outweigh the benefits. Patients who may currently only visit their clinic once or twice a year will need to come in six or 12 times a year, which could be inconvenient. The need to take time away from work or personal responsibilities could lead to unwanted disclosure of HIV status for some. Some people dislike injections or the discomfort they can cause. Many people living with HIV feel comfortable and confident taking pills every day, and would still need to have daily medication for other health conditions even if their HIV treatment was less frequent. Some people feel uncomfortable with the idea of having an active medication in their body for several weeks or months at a time, and being unable to change that once it has been administered.
People who have taken injectable treatment as part of research studies have generally been very satisfied with it. Some more ambivalent views have been expressed by other people with HIV in studies in France and the United States.
Does injectable treatment help with adherence?
We don’t know yet, but long-acting technologies are already used for other health issues, such as contraception, osteoporosis, and psychiatric treatment. This experience suggests that long-acting medications may help people miss fewer doses and it is plausible that this could also be true for HIV. However, the studies which led drug regulators to approve cabotegravir and rilpivirine only gave the injections to people who had already had good adherence to daily pills.
Since injectable cabotegravir and rilpivirine were approved, an HIV clinic in San Francisco has been providing injectable treatment to people with detectable viral loads through a special programme which combines treatment with intensive follow-up support. The programme has shown promising results, with 55 out of 57 patients maintaining an undetectable viral load after a year and a half, despite many having never achieved viral suppression before.
Another larger US study found injections with cabotegravir and rilpivirine monthly or every other month can be an effective option for people who have difficulty maintaining viral suppression because they find adherence to daily pills challenging. In the first phase of this study, people continued with daily oral therapy but received comprehensive adherence support and financial incentives to achieve viral suppression. If they were able to do so, they were eligible to switch to the injectable regimen.
Rather than adherence to daily pills, cabotegravir and rilpivirine require adherence to healthcare appointments once a month or once every two months. We don’t yet know how well people will stick to this when injections are provided in regular healthcare settings, rather than in research studies.
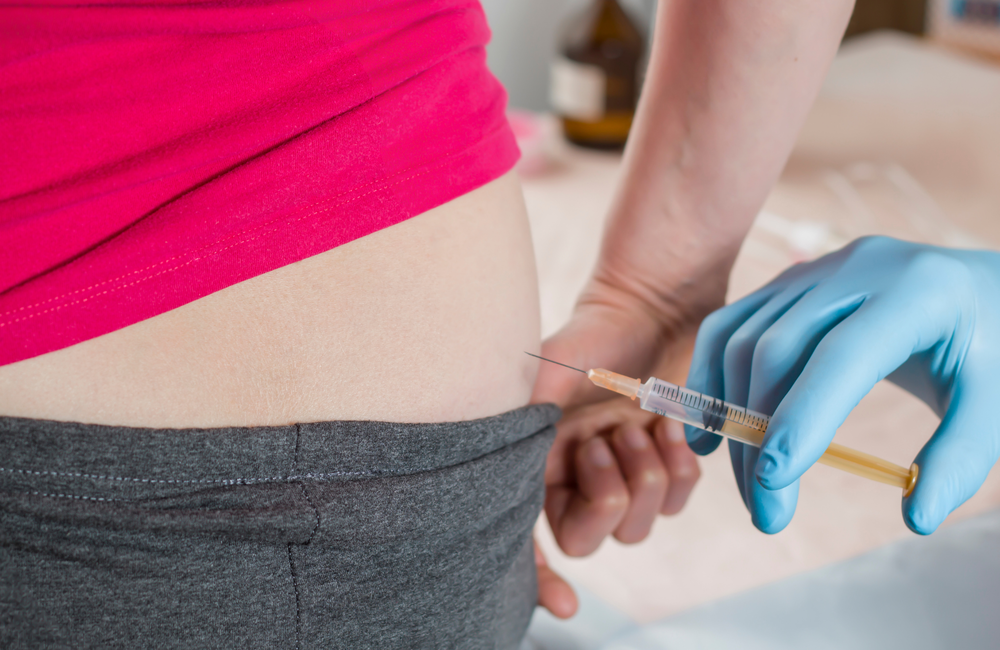
Where are the injections given?
Both injections are given into the muscle of the buttocks, a few minutes apart, by a healthcare professional. As cabotegravir and rilpivirine must be injected into a muscle large enough to take the required volume of medication, the buttocks are the ideal injection site.
Studies are being done to see whether injections in other parts of the body, such as the thighs, are safe and effective. If successful, self-injection might become an option.
How frequent are the injections?
Injectable cabotegravir and rilpivirine can be taken either once a month or every two months.
When taking standard HIV treatment, you need to take your tablets every 24 hours to have enough levels of the medications in your blood. Injectable cabotegravir and rilpivirine have long half-lives, meaning they can stay in the body for much longer than the tablets. This makes long-acting treatment possible: injections every one or two months are enough to maintain high enough blood levels of the medication to suppress HIV.
Your doctor will tell you whether you need to attend each month, or every two months, for your injections. The frequency of injections may depend on your country. For example, the pharmaceutical company has decided that in Europe it will only sell the dosage required for the every-two-months regimen. (Depending on whether the injection is every one or two months, different doses are given.)
What are the side effects?
Around three-quarters of people who take cabotegravir and rilpivirine have injection site reactions such as pain, bumps and swelling. They typically last a few days and tend to become easier to tolerate after the first few injections. Very few participants dropped out of the studies due to these reactions.
Other common side effects are headache, raised temperature and feeling hot.
There’s more information about side effects in our factsheet and in our detailed page in the A-Z of antiretroviral medications.
Is injectable treatment safe to use during pregnancy?
Injectable cabotegravir and rilpivirine is not currently approved for use in pregnancy or while breastfeeding. This is because pregnant and breastfeeding people were not included in the studies used to approve the drugs, so doctors are not sure about their potential effects on the risk of miscarriage or birth defects. A clinical trial of injectable cabotegravir and rilpivirine in pregnancy is currently underway.
Is injectable treatment safe to use with other medications and gender-affirming hormones?
Injectable cabotegravir and rilpivirine should not be used with:
- the anticonvulsants carbamazepine, oxcarbazepine, phenobarbital, phenytoin
- the antimycobacterials rifabutin, rifampicin, rifapentine
- the systemic glucocorticoid dexamethasone, except as a single-dose treatment
- the herbal supplement St John’s wort.
Injectable cabotegravir and rilpivirine are safe to use with gender-affirming hormones, and also with contraception.
Injectable cabotegravir and rilpivirine has fewer drug interactions than older HIV drugs. As the medications are absorbed by the body in different ways to oral medications, some drug interactions are avoided.
However, as it is more widely used, it is possible that some unexpected drug interactions may occur. These may be difficult to manage because it is not possible to quickly remove cabotegravir and rilpivirine from the body once they have been injected. Therefore, consultation with a pharmacist is important both before you start injectable treatment, and, if currently taking it, before taking other prescription medicines, over-the-counter medicines and supplements.
There’s more information about drug interactions in our factsheet and in our detailed page in the A-Z of antiretroviral medications.
Is it necessary to take daily tablets before having the injections?
It was initially recommended to start by taking cabotegravir and rilpivirine pills once a day for a month, before beginning the injections. This is known as an ‘oral lead-in’.
The rationale is to ensure that the medications are well tolerated since the content of an injection cannot be removed if it causes problems. Nonetheless, safety concerns have not emerged from the trials so far, and there are some data suggesting the oral lead-in is not necessary. As a result, regulators have now approved the oral lead-in as optional, and you can decide with your doctor if this is something you want to do.
How long do the drugs stay in the body after an injection?
The injectable medications persist in the body for several weeks or months after a last injection, slowly declining over time. It is estimated that the ‘half-life’ (how long it takes for the drug concentration in blood to be reduced by 50%) of cabotegravir is between five and 12 weeks, while that of rilpivirine is between 13 and 28 weeks. In some people, traces of the drugs can still be found one year after their last injection.
This raises concerns about drug resistance. If you stop receiving the injections and do not switch to another HIV treatment, there will be a lengthy period during which resistance could develop. Drug resistance can develop in situations like this when there is some HIV medication in the blood but not enough to fully suppress the virus.
For this reason, if you stop the injections, it is essential to switch to an alternative HIV treatment regimen. If you have been having the injections once a month, you should switch to your new treatment within one month of your final injection. If you have been having them once every two months, you should switch within two months.
What happens if you are late for an injection?
The injections do require people to attend injection appointments regularly. Nonetheless, there is a 14-day dosing window – it’s OK to have the injection up to seven days before or up to seven days after the regular date. The 14-day window applies to both the monthly and every-over-month schedule, but having the injections on time is particularly important for those using the every-other-month regimen.
If you know you won’t be able to attend an injection appointment (for example, if you are travelling), you need to let your clinic know. You will be given a supply of cabotegravir and rilpivirine tablets to take each day until you can go back for more injections. It is important to take these tablets as prescribed as otherwise there is a risk of developing drug resistance if you have your injection outside of the dosing window.
If you miss an injection or it is delayed by more than seven days and you have not taken pills during the gap, your doctor will reassess whether injectable treatment is the right option for you.
How will health services adapt?
Arrangements for providing the injections are likely to vary considerably between countries, due to different health systems, policy challenges and funding arrangements. In many places, the injections will probably be initially provided at specialist HIV services; it may take some time for more convenient community-based services to be put into place. Implementation studies in the US, UK and Europe are currently evaluating delivery of injections at home and at HIV community-based organisations to see if they are workable treatment options.
HIV services will need to make some adjustments to their clinics. Patients who may currently only visit their clinic twice a year will be attending six or 12 times a year. Administration of injections is resource-intensive, requiring more staff time and clinic space than oral therapy. Clinics must establish a system to schedule and track injection appointments, send reminders, and trigger intervention for people who are late for a dose. Staff need to be trained in the injection technique and on how to support patients.
You can read about early adopters’ experience implementing injections in our news reports:
- Long-acting injectable HIV treatment is feasible, even during COVID-19
- Clinics will need new systems to administer injectable HIV therapies, says Canadian early adopter
- Injectable HIV medications must be practical and affordable, conference hears.
Will injectable HIV treatment be practical in low- and middle-income countries?
So far, long-acting antiretrovirals have mostly been studied in men from higher-income countries, which is not representative of the global HIV epidemic. Research in more diverse settings is needed.
The cost of cabotegravir and rilpivirine could be a significant barrier to provision in low- and middle-income countries. Highly effective and safe oral antiretroviral therapies already exist, at a cost of less than US$50 a year, and the injectables’ price will need to be competitive against this. While generic manufacturers have plans to produce low-cost cabotegravir for PrEP, it’s not clear whether they will also produce rilpivirine. In addition, the work processes of already over-burdened clinics will need to be rethought, or alternative service providers identified.
Several African countries do already have large programmes providing contraceptive injections, suggesting it may be feasible to administer injectable HIV treatment too. However, there may be extra challenges. Rilpivirine needs to be stored in a fridge (whereas the contraceptive injection does not); cabotegravir and rilpivirine is not suitable for people with HIV and hepatitis B co-infection (which is common in many African and Asian countries); and they should not be used at the same time as some tuberculosis medications (rifampicin and rifapentine).
According to a discussion at an international conference in 2023, injectable treatment will likely only be available in low- and middle-income countries via small-scale, demonstration projects for the foreseeable future.
Will other injectable medications be available in the future?
Developing new long-acting medications is currently a major focus of pharmaceutical companies’ research. As many of the daily pills for HIV treatment are highly effective and well tolerated, creating simpler treatment options is seen as the main way to make progress. Various companies are trying to develop intramuscular injections, subcutaneous injections, infusions, implants, patches and pills.
This process is not smooth, with many potential products failing for one reason or another. For example, several clinical trials of islatravir, which had been considered to be one of the most promising new antiretrovirals, were halted in December 2021 due to declines in immune system cells in some people taking the medication. While research has restarted on a weekly islatravir pill at a lower dose combined with a six-monthly injection of lenacapavir, as well as trials of daily islatravir combined with other pills, development of a monthly pill and an annual implant has been shelved.
Several broadly neutralising antibodies (bNAbs) are being studied for HIV treatment, prevention and long-term viral remission. One phase II study is pairing VRC07-523LS (a bNAb, given as an infusion every two months) with cabotegravir (given as an injection every month). Another is pairing 3BNC117 (a bNAb, given as an infusion every two or four weeks) with albuvirtide (a fusion inhibitor, given as a weekly injection).
Other medications in development include leronlimab (PRO 140, a CCR5 antagonist, given as a weekly subcutaneous injection) and UB-421 (a CD4 attachment inhibitor, given as an infusion every two weeks). However, some researchers have questioned whether bNAbs will offer a viable alternative to ART if their performance does not improve.
Scientists have also identified a way of creating a monthly injectable version of the world’s most commonly prescribed antiretroviral combination, tenofovir disoproxil fumarate, lamivudine and dolutegravir (also known as TLD), using nanoparticle technology. This could offer a more affordable treatment option due to the low production costs of the three drugs. However, it is yet to be tested in humans.
Although lenacapavir has already been approved for use in people with multi-drug resistant HIV, its promise as a long-acting treatment has so far been limited as it needs to be taken with a second medication (usually daily pills). A new study which combined injectable lenacapavir with a pair of bNAbs (teropavimab and zinlirvimab, given as infusions) every 6 months has shown promising results. A phase II trial of this treatment regimen has begun to see whether viral suppression can be maintained over a longer period of time in a larger group of people.
