Key points
- This is one type of long-acting PrEP – an injection taken to prevent HIV infection.
- It’s often known by its brand name, Apretude.
- This page provides detailed information about how to start and stop PrEP, required tests and checkups, side effects, drug interactions and drug resistance.
- Other types of PrEP are also available.
What is PrEP?
PrEP stand for pre-exposure prophylaxis. This means preventing HIV by taking anti-HIV drugs.
PrEP allows you to have the sex you want without the fear of contracting HIV.
How does it work?
The anti-HIV drug in PrEP stops HIV from entering your cells. You need to have injections every two months to make sure that the drugs maintain high levels in your bloodstream to prevent HIV infection.
What is cabotegravir?
Cabotegravir is a long-acting form of PrEP, taken by injection once every two months. Its brand name is Apretude. Healthcare professionals sometimes call it long-acting cabotegravir or CAB-LA.
The same medication can also be used by people living with HIV as part of their treatment, in combination with another injected medication. This is different from PrEP. There’s more information about this on another page.
What other types of PrEP are there?
The most widely used form of PrEP is a tablet containing tenofovir disoproxil / emtricitabine, and there’s also an alternative PrEP tablet containing tenofovir alafenamide / emtricitabine.
An alternative PrEP injection contains lenacapavir and is taken every six months, and there’s also a vaginal ring containing dapivirine. However, not all the types of PrEP are available in all countries.
What’s the difference between PrEP and PEP?
You start to take pre-exposure prophylaxis (PrEP) before you are exposed to HIV and continue to take it for as long as you need it, to protect against infection. You take post-exposure prophylaxis (PEP) after you have been exposed to HIV, for a fixed period of one month.
Effectiveness
In studies, injectable cabotegravir used as PrEP was more effective than oral PrEP consisting of tenofovir disoproxil and emtricitabine because it is more difficult to miss doses. In a large study in gay and bisexual men and transgender women, injectable cabotegravir PrEP reduced the risk of acquiring HIV by 66% compared to oral PrEP. In a large study in cisgender women, injectable cabotegravir PrEP reduced the risk of acquiring HIV by 88% compared to oral PrEP. The risk reduction could have been higher in women because missing doses of oral PrEP may have a bigger impact on how well PrEP works in women than in men.
Is this type of PrEP available to me?
At the time of writing, cabotegravir has been approved by drug regulators in over 60 countries in all parts of the world. You can check the PrEPWatch website to see if it approved for use in your country.
In many of countries where cabotegravir has been approved by drug regulators, the injections are not yet available in clinics and decisions on funding and reimbursement still need to be made. As this type of PrEP is more expensive than PrEP tablets, its use may be restricted to individuals who have difficulties taking tablets regularly.
Services which provide HIV testing, treatment for sexually transmitted infections or treatment for HIV may also provide PrEP – or have information on local services which do.
Tests before starting PrEP
You need to have an HIV antibody / antigen test to check that you don’t have HIV already. If you have HIV, taking PrEP will lead to your HIV developing resistance to cabotegravir. As this type of drug is also commonly used in HIV treatment, it’s very important to avoid this.
How to take it
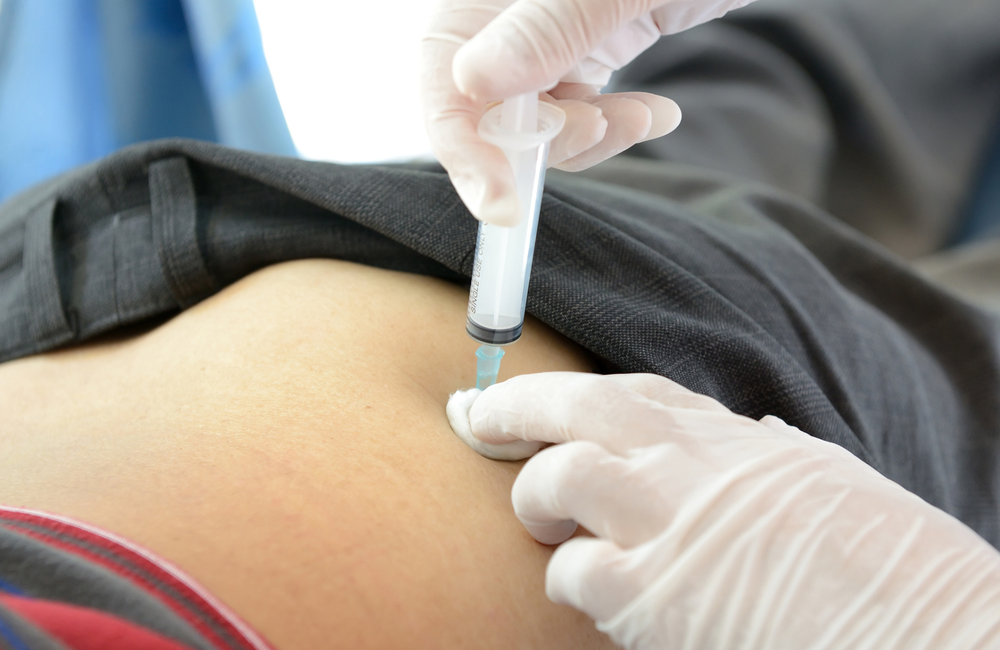
Cabotegravir PrEP is given as an injection, into the muscle of your buttock, by a healthcare professional.
For it to work, you need to have enough PrEP in your body when you need protection. It’s therefore important to attend your clinic as scheduled in order to receive your injections on time. The injections are usually every two months, but the schedule is a little different at the beginning.
As PrEP may stay in the body for several months after the last injection, there are some procedures you need to follow if you decide to stop taking it. (See below, under ‘Stopping PrEP’.)
Starting PrEP
There are two different ways you can start:
- You can start with one injection, followed by a second injection one month later. After the second injection, you will receive cabotegravir injections every two months.
- Alternatively, you take cabotegravir as a daily tablet for one month before starting the injections. This means that if you are unable to tolerate cabotegravir, you can stop taking it and the side effects will go away quickly.
If you start taking cabotegravir PrEP as tablets, take one tablet each day at the same time of day. You can take cabotegravir tablets with or without food. Take cabotegravir tablets for at least 28 days before your first injection.
One week after the first injection, cabotegravir will provide complete protection against HIV.
Continuing PrEP
Injections are every two months. There is a 14-day dosing window – it’s OK to have the injection up to seven days before or up to seven days after the regular date.
If you know you won’t be able to attend an injection appointment (for example, if you are travelling), you need to let your clinic know. You may be given a supply of cabotegravir tablets to take each day until you can go back for more injections.
Stopping PrEP
If you stop taking cabotegravir injections (and are not taking cabotegravir tablets), cabotegravir levels in your body will decline slowly and small amounts of the drug will still be present for several months. One year after their last injection, traces of the drug are still present in around one in five people.
If you acquire HIV during the period between stopping cabotegravir PrEP and all the cabotegravir leaving your body, there is a risk that the HIV you acquire will develop resistance to cabotegravir. As this would mean that cabotegravir and other similar drugs would not work properly as HIV treatment, it’s important to avoid this.
You should not assume that injectable cabotegravir will continue to provide protection against HIV more than two months after your last injection. If you still need protection from HIV, it is important to take other precautions from this point. You could take daily oral PrEP tablets for 12 months after stopping cabotegravir injections to avoid the risk of acquiring HIV that is resistant to cabotegravir.
It's also important to go back to your clinic for HIV tests every three months.
If you have stopped taking PrEP and have had sex that could have exposed you to HIV, contact your clinic without delay to see if you should take a 28-day course of post-exposure prophylaxis (PEP). It’s best to start this within 24 hours, and definitely within 72 hours. There’s more information about PEP on another page.
Checkups while taking PrEP
Regular checkups are important while taking PrEP.
You will be tested for HIV at the time of your first injection and every subsequent injection, to check that you remain HIV negative. You should be tested for HIV with a fourth-generation antibody-antigen test and a viral load test. A fourth-generation antibody-antigen test is more sensitive than a rapid antibody test and will detect HIV sooner. The viral load test is used to detect HIV before your body has produced antibodies against HIV, during the first weeks of infection.
If you stop taking injectable cabotegravir PrEP and subsequently have condomless sex more than two months later, you will need to get a fourth-generation antibody test before starting PrEP again.
If you are not taking PrEP and you have flu-like symptoms, possibly with a rash, within a month of condomless sex, this may be a symptom of recent HIV infection. You should have a fourth-generation HIV test a month after the most recent exposure and delay starting or re-starting PrEP until you have been confirmed HIV negative.
If you have just started PrEP and you have flu-like symptoms, possibly with a rash, this may be a symptom of recent HIV infection. Contact your clinic immediately to arrange a fourth-generation HIV test and/or viral load test. Your clinic may add other drugs to your PrEP in case you have HIV.
Telling your GP you are taking PrEP
It is a good idea to tell your general practitioner or primary care physician that you are taking PrEP containing cabotegravir, to avoid interactions with other prescription medicines.
Using PrEP during pregnancy, breastfeeding or when you are trying to conceive
The available data suggests that cabotegravir injections are safe to use during pregnancy and breastfeeding, and when trying to become pregnant.
Who shouldn’t take PrEP?
People with HIV shouldn’t take injectable cabotegravir due to the risk of developing of resistance. This is why it is important to test for HIV before starting cabotegravir PrEP.
Side effects
The most common side effects of PrEP containing cabotegravir (affecting at least one in ten people) are:
- Injection site reactions (pain, tenderness or lumps at the injection site)
- Diarrhoea
- Headache
- Raised liver enzymes
- Feeling hot or feverish.
Many people find they get more used to injection site reactions after a few injections. For the first two or three injections, you could take an over-the-counter pain medication within a couple of hours of the injection and continue as needed for one to two days. You can also apply a warm compress or heating pad to the injection site for 15-20 minutes.
Other common side effects (affecting at least one in a hundred people):
- Difficulty sleeping, abnormal dreams, depression, anxiety, dizziness
- Feeling sick, abdominal pain, flatulence
- Rash
- Muscle pain
- Tiredness, weakness.
Some uncommon but serious side effects (less than one in a hundred) are:
- Suicidal thoughts or behaviours (people with a pre-existing psychiatric illness may be more likely to experience).
- Hypersensitivity or allergic reaction
- Liver inflammation (hepatotoxicity).
If you have side effects which don’t go away or get worse, speak to staff at your clinic.
Interactions with other medicines
Do not take cabotegravir PrEP while using the following medicines:
- Carbamazepine, oxcarbazepine or phenobarbital (anti-convulsant treatments)
- Phenytoin (epilepsy treatment)
- Rifampicin or rifapentine (antibiotics used to treat tuberculosis).
PrEP does not interact with:
- Alcohol or recreational drugs
- Hormonal contraception
- Gender-affirming hormone therapy
- Erectile dysfunction drugs
- Anabolic steroids.
The University of Liverpool provides an online tool to check for interactions between anti-HIV drugs (including PrEP) and other medications. You enter the names of your PrEP drugs (tenofovir disoproxil and emtricitabine) in the left column, and the other medications you are taking in the next column. The results are provided with a traffic-light system: if the result is red or amber, it’s worth checking with your doctor or pharmacist. If it’s green, there shouldn’t be any problem. Visit www.hiv-druginteractions.org/checker or download the Liverpool HIV iChart app for iPhone or Android.
Drug resistance
Some people worry that while taking PrEP they may get drug-resistant HIV. It’s important to remember that HIV drug resistance can only happen if you take PrEP when you already have HIV. This is why you should get tested for HIV before you start PrEP, and keep testing regularly.
If you continue taking PrEP or start taking PrEP again after you acquire HIV, this could drive HIV to develop resistance to the drug you are taking as PrEP. If you acquired HIV in the 12 months after stopping PrEP, you could also develop resistance – this is because traces of cabotegravir can remain in the body for up to a year after your last injection.
Resistance to cabotegravir and other drugs of this type (integrase inhibitors) would restrict your options for treating HIV.
There is another way in which resistance can affect PrEP. There are a very small number of people who have acquired HIV when taking PrEP despite having adequate drug levels in their blood. Some of them may have acquired a form of HIV that had resistance to the PrEP drug cabotegravir.
There’s more information about resistance on another page.
Does PrEP prevent STIs?
Injectable cabotegravir PrEP does not prevent other sexually transmitted infections so it’s a good idea to have regular tests for sexually transmitted infections while you are taking PrEP.
