Long-acting injectable cabotegravir/rilpivirine
The development of medications for HIV therapy that do not need to be taken every day has been a major theme of the HIV Glasgow 2020 conference, held virtually last week. Several presentations reported on injectable cabotegravir/rilpivirine, the long-acting medication which is the closest to regulatory approval.
Cabotegravir is an experimental integrase inhibitor and rilpivirine is a non-nucleoside reverse transcriptase inhibitor (currently available in pill form as Edurant). Data from the FLAIR study in people taking antiretroviral therapy (ART) for the first time has already showed that more than 90% of people receiving monthly injections have an undetectable viral load after 96 weeks of treatment.
At HIV Glasgow, Professor Chloe Orkin of Queen Mary University of London presented further results showing that cabotegravir/rilpivirine was effective regardless of whether study participants started the injections immediately or took oral versions of the drugs for four weeks before switching to injections.
The rationale for the oral lead-in was to ensure that the medications are well tolerated, since the long-acting injectables cannot be removed if they cause problems. However, there were no specific safety concerns in the earlier part of the study and other long-acting injected medications, such as psychiatric drugs, do not require an oral lead-in.
After 96 weeks, FLAIR participants who had originally been randomised to receive oral ART were offered cabotegravir/rilpivirine. They could choose to receive the injections immediately or to start with the four-week oral lead-in. Although allocation was not randomised, most patient characteristics were comparable between the 111 people who received the injections immediately and the 121 people who had the oral lead-in.
Twenty-four weeks later, 99.1% and 93.4% of the two groups respectively had an undetectable viral load. A pharmacokinetic analysis found that drug concentrations in the body did not differ based on whether or not the oral lead-in was used. Treatment was generally safe and well tolerated. About one in five people in each group experienced drug-related adverse events, but there was only one severe event, which was not judged to be treatment-related.
Uptake and cost-effectiveness of injectables
Two-thirds of people on HIV treatment would like to switch to a long-acting injectable regimen, according to a 688-participant survey conducted in France, Germany, Italy and the United Kingdom.
Patients who were interested were more likely to be younger, more recently diagnosed, heterosexual and born overseas. People with specific unmet needs generally agreed that the injectable would help with those issues. This included people who feel anxious about missing a dose, people who dislike the daily reminder of their HIV status, individuals with suboptimal adherence and people with confidentiality or privacy concerns (such as worrying that taking pills every day increases the chance of their HIV status being revealed).
The researchers also surveyed 120 clinicians. They were generally open to offering long-acting injectables to patients, assuming that they were not more expensive. Nonetheless, the clinicians only thought that 26% of patients would actually switch.
The survey was run on behalf of ViiV Healthcare, who make injectable cabotegravir/rilpivirine, so it might be expected to come up with a favourable result. However, an independently run Italian survey found an even greater level of interest, with 89% of respondents being interested in injectables.
Another analysis presented by ViiV covered cost-effectiveness. It found that, if injectable cabotegravir/rilpivirine improves adherence relative to oral drugs, it should save the money for the healthcare system, at least over a patient's lifetime. It would also add a few weeks to the time people with HIV who take it can expect to live in good health and avert some HIV transmissions.
The researchers assumed that there is no difference in the biological efficacy of injectable and oral treatment, and that the annual cost of the injectable therapy would be the same as the average cost of the three most widely used oral therapies.
However, adherence was expected to be different. Attendance at clinic appointments for injections was assumed to be at the same excellent level as has been seen in clinical trials. In contrast, adherence to oral therapies was assumed to be between 5 and 25% lower than for injections.
This poorer adherence to oral therapies was projected to result in some people developing detectable viral loads and low CD4 counts and eventually needing more expensive therapies. Assuming 5% poorer adherence to oral therapies, using the injectables in 1000 patients would result in an overall lifetime saving of treatment costs of £4.2 million. There would be an increase in time in good health of 43 years per 1000 patients, which averages out to just over two weeks per patient. Due to periods of unsuppressed viral loads, there would be two extra HIV transmissions per 1000 patients if adherence to oral therapies was 95%.
Greater cost savings would be seen at lower levels of adherence to oral therapies.
New oral option that might be dosed once a week
The conference heard results of dose-finding studies of a new drug, MK-8507, in the non-nucleoside reverse transcriptase inhibitor (NNRTI) class, which suggest that it could be given just once a week.
Initial studies were conducted in HIV-negative volunteers, which helped select doses for further study and demonstrated that drug absorption was not affected by food.
The next tests were done in HIV-positive volunteers who had not previously taken antiretroviral therapy. Doses of either 80mg or 300mg both produced a similar drop in viral load of about 1.5 logs, which equates to a 35-fold reduction.
Levels of MK-8507 stayed above the therapeutic levels for just under a week on the 80mg dose, and for two weeks on the 300mg dose.
A 40mg dose was not as long-acting or as potent.
The drug’s manufacturer Merck & Co already has another investigational drug, islatravir, which can be dosed once a week. They are planning future studies in which an oral combination of islatravir and MK-8507 (80mg) is provided once a week.
Concerning islatravir, a separate presentation at HIV Glasgow provided 96-week results from a phase IIb study in people starting HIV treatment for the first time. It evaluated the safety and efficacy of daily oral islatravir plus the NNRTI doravirine, in comparison with a three-drug regimen of doravirine, tenofovir disoproxil fumarate and lamivudine, the components of the Delstrigo single-tablet regimen.
At week 96 after starting treatment, 90% of people treated with the selected dose of islatravir plus doravirine had undetectable HIV viral load, showing that the combination works at least as well as a standard three-drug regimen. Treatment was generally safe and well tolerated.
Islatravir is now undergoing testing in a comprehensive clinical development programme. This includes phase III studies of people starting treatment, heavily treatment-experienced people and people with viral suppression who are switching from other regimens. A phase II study of children and adolescents is also planned.
In addition, islatravir is being studied for pre-exposure prophylaxis and post-exposure prophylaxis (possibly using weekly or monthly doses). Early studies also suggest that a long-acting islatravir implant could provide protection for a year.
The impact of COVID-19 on HIV services in Europe
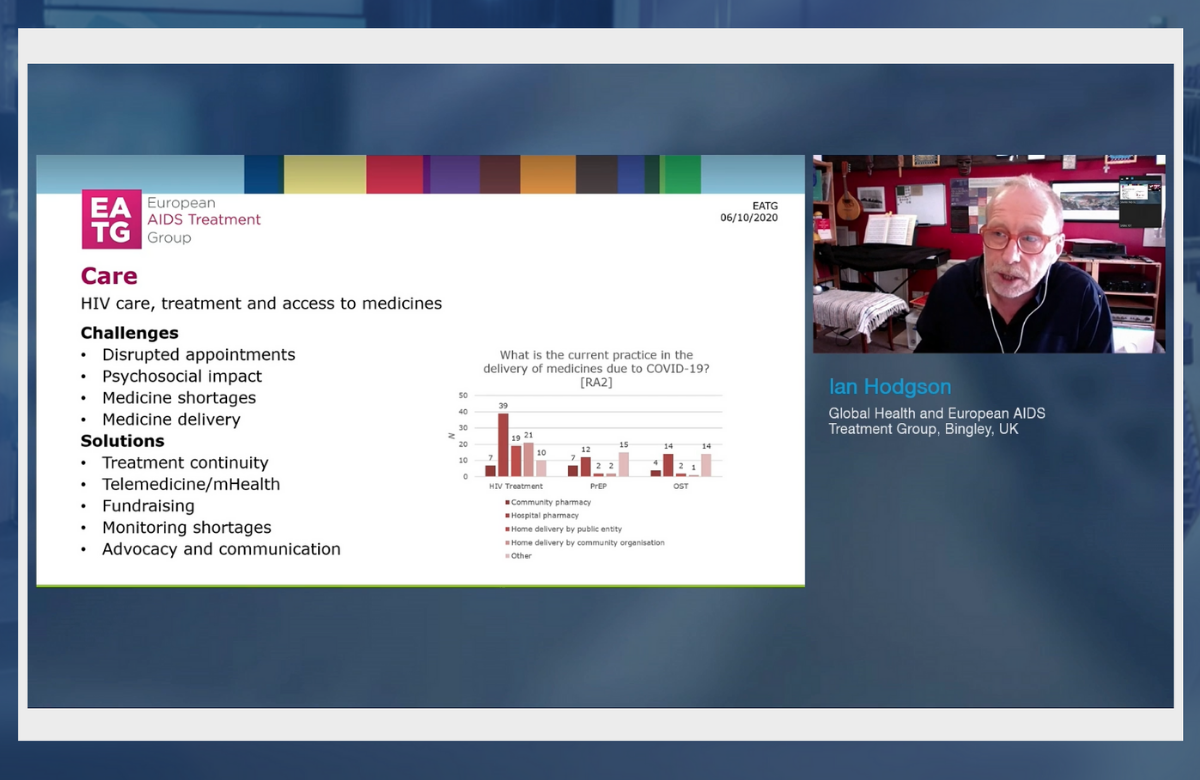
A series of rapid-response surveys conducted by the European AIDS Treatment Group (EATG) has uncovered significant changes in access to HIV testing, PrEP and HIV treatment during the course of the COVID-19 epidemic. The findings do not just document negative impacts, but also show that a number of community organisations have found innovative solutions to these.
HIV testing seemed to be more disrupted in countries with stricter anti-COVID measures, with rapid and outreach HIV testing services often suspended altogether. However, in many places there had been an increase, or at least continued availability, of self-testing, with many NGOs putting on campaigns to increase awareness of self-testing. Half of respondents said that sexually transmitted infection testing in their area was only available for acute infections and emergencies, not for routine tests.
Respondents from nearly all countries reported disruption to normal HIV appointments, which were either postponed, with extra supplies of antiretroviral therapy (ART) being sent, or appointments being provided on the phone. In a number of countries, the diversion of physicians into COVID care had left a severely inadequate service.
There was evidence that community and healthcare organisations were responding to the pandemic with innovative solutions. These ranged from extended prescriptions to community-based organisations providing more access points when the usual peripheral clinics had been closed. Healthcare workers were taking drug delivery into their own hands and taking medication to patients by car or bicycle. Community pharmacies had been opened or given permission to dispense ART.
Ian Hodgson of EATG told the conference that the challenge now was for community organisations to change from an emergency to a strategic response to the COVID pandemic. Funders’ priorities might change permanently, he said, and organisations might need to change their aims and priorities to address the needs of the post-COVID world.
Women’s involvement in clinical trials
Women’s under-representation in clinical trials is motivated by a desire to protect them and their unborn children from potential harm caused by a medication, but this results in inequity for women: unknown safety, efficacy and tolerance of most new medications. A session at HIV Glasgow explored the biological, clinical and ethical imperatives for involving diverse women in HIV clinical trials.
There are biological differences which have clinical implications, Dr Catherine Orrell told the conference. In general, women weigh less and have a higher percentage of body fat than men, which can alter the pharmacokinetics of a drug in their body. Likewise, hormonal changes occurring throughout the menstrual cycle and in pregnancy have an impact on how drugs are processed in the body. Drugs may have a different effect in men and women: for example, the antiretroviral nevirapine induces hepatitis and rash at a much lower CD4 count in women than in men.
The association of dolutegravir with weight gain in women only emerged after the drug had been approved by regulators (weight gain had not even been recorded in the initial clinical trials). Similarly, dolutegravir’s possible association with neural tube defects only emerged after approval, and policymakers had to rely on one major study for data on the link, with a lack of other sources of data to provide a more comprehensive picture.
While there are appropriate ethical concerns about harming the unborn child, this ignores the reality that medications will inevitably be used by pregnant women after regulatory approval, especially as half of pregnancies are unplanned. It’s therefore preferable to find out about any risks in the controlled environment of a clinical trial, with appropriate precautions and regulations, than for the drug to be used in everyday clinical practice without that information, Professor Maggie Little argued.
Pregnant women living with HIV should not be protected ‘from’ clinical research, but protected ‘through’ research, she said. Women and their doctors need to have reliable data on dosing adjustments, foetal safety and maternal safety.
Activist Longret Kwardem said that women want to be meaningfully engaged in research from the start to the end. They also want women-specific research through the life course, with consideration for their mental health and quality of life.
Weight gain on HIV treatment
Delivering a keynote lecture, Professor Andrew Carr of St Vincent’s Hospital, Sydney, urged caution in thinking about changes in body fat, pointing out that younger HIV-negative adults in the United States gain up to 1kg a year. Large studies have reported that over several years, people with HIV on antiretroviral treatment gain weight – but their weight gain only returns them to levels that are typically seen in the societies where they live.
Several large trials have shown that the median weight gain in people starting integrase inhibitor treatment is in line with average annual weight gain for the population. Mean weight gain, on the other hand, can be much higher in these studies, distorted by a minority of outliers who have the greatest weight gain.
However, clinicians should be watching out for the most extreme cases, as these individuals are most likely to suffer weight-related complications in the future. General population studies show that a 5kg/m2 increase in body mass index significantly increases the risk of death. “There’s no biological reason to believe that obesity in HIV-positive adults will not incur the same risk as it does in the general population,” said Carr.
Reminding delegates that it took over five years to show that lipoatrophy could be prevented by avoiding thymidine nucleoside analogues in randomised trials, he appealed to researchers to help answer questions about causes of weight gain by designing studies that could tease out the contributions of individual drugs. Comparisons between sexes, ages and races are also needed.
Studies of people changing antiretroviral regimen or taking PrEP are more likely to identify drug effects. Studies in previously untreated people may show ‘return to health effects’ and interpreting weight changes is complicated by the fact that drugs are given in combination.
In the same session, Dr Giovanni Guaraldi of the University of Modena, Italy, said that fat tissue distribution and density is more important than total fat gain in determining the risk of cardiovascular disease and metabolic disorders. People with HIV who gain weight after switching to an integrase inhibitor are not gaining a disproportionate amount of visceral fat which is linked to metabolic disorders. Instead, the gain in body mass was driven by 'good fat', said Guaraldi.
A sleep intervention

Many people living with HIV complain of disturbed sleep. UK clinicians reported that a questionnaire on sleep quality, validated in other disease areas, appears to be reliable, simple and convenient for assessing sleep disturbance in people with HIV.
Patients assessed as having disturbed sleep were offered information on sleep hygiene and given the option to switch antiretrovirals if appropriate. Sleep quality improved after these interventions, especially among those patients who chose to switch from dolutegravir or another antiretroviral.
NAM aidsmap conference bulletin survey

We would love to find out what you think about our conference bulletins, especially those that you’ve received in the last year. Could you please help us by completing this short survey? We will use the feedback to report back to our funders. The survey will take no more than 5-10 minutes to complete.
