Impact of COVID-19 on sexual behaviour and PrEP use
These were the findings of several late-breaking studies presented to the 23rd International AIDS Conference (AIDS 2020: Virtual), taking place online this week.
A cohort of 940 gay and bisexual men in Australia were asked in April about sexual behaviour pre- and post-COVID-19. Participants reported a 65% reduction in sex with recurring but casual partners and a 58% reduction in sex with casual partners. Prior to COVID-19, 46% had been on PrEP and 58% continued to use it. Of those stopping PrEP, 86% gave COVID-19 as one of the reasons, but only 17% reported that they had found it difficult to access PrEP during this time.
A survey of 394 PrEP users and 188 healthcare providers across the US demonstrated similar themes of behavioural change and reduction of PrEP uptake, generally by choice rather than restricted access. The 95.3% of users on daily PrEP went down to 61.6% while the much smaller group using event-driven PrEP dropped from 4.7% to 2.9%. These reductions were mirrored by reported reductions in risk activities.
Data from PrEP providers showed over 90% restricting or changing their practice to reduce face-to-face contact, but many reported introducing telephone or online appointments to triage patients. Only 2.6% had closed entirely and stopped offering PrEP in any way.
Difficulty in accessing services from labs made it hard for providers to follow national PrEP guidelines. Almost half had treated patients for suspected sexually transmitted infections without testing.
A review of the impact of COVID-19 on PrEP services at an LGBT+ health facility in Boston confirmed these findings and highlighted impacts on vulnerable sub-populations.
Dr Douglas Krakower said that his examination of 3520 patient records from January to April showed the number of new patients starting PrEP reduced by almost three-quarters over the period. Prescription lapses (where no new PrEP prescription was filled before a previous one ran out) rose from February onwards and occurred more often in younger people, black and multiracial people, trans people and people with public health insurance.
New York studies on COVID-19 in people with HIV
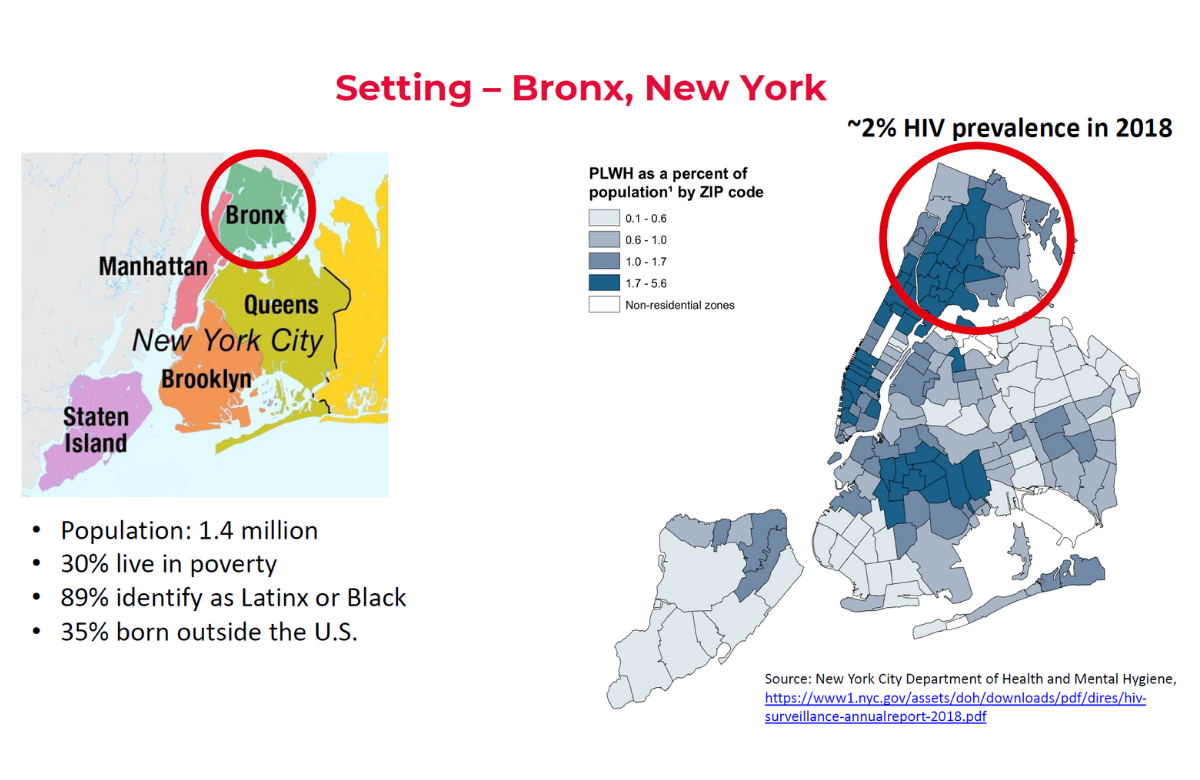
The retrospective cohort study focused on 100 HIV-positive and 4513 HIV-negative people who were hospitalised between March and May in the Bronx, one of the epicentres of the New York City COVID-19 outbreak. Just over half were men and the median ages were 62 and 65 years, respectively. Approximately 40% were black, a similar proportion were Latino and 6% were white. Most were on Medicaid (coverage for people with low income) or Medicare (coverage for older people), with only about 15% having private health insurance. Among the people living with HIV, 81% had a suppressed viral load, however, about one in five had a CD4 count below 200.
Most outcomes did not differ between the HIV-positive and HIV-negative patients, including length of stay in hospital (five days in both groups), rates of acute kidney injury or likelihood of death while hospitalised (22% vs 24%, respectively). People with HIV did have a higher rate of intubation and mechanical ventilation, but the difference fell just short of statistical significance. Unexpectedly, the study found that the likelihood of intubation and death in hospital was higher for people with viral suppression compared to those with uncontrolled HIV.
A separate study looked at clinical and immunological characteristics in 93 HIV-positive people with COVID-19 attending five emergency departments in New York in March and April. This study had no HIV-negative control group for comparison.
It found that people with HIV had higher levels of inflammation and cytokines, indicating that they are capable of mounting a strong inflammatory response to the new coronavirus and remain at risk for severe COVID-19 despite taking antiretroviral therapy.
Dr John Hsi-en Ho noted that coronavirus testing was limited during this period and tended to be reserved for sicker patients, so this group is unlikely to be representative of all HIV-positive people with COVID-19.

COVID-19 speeds up implementation of multi-month dispensing
Multi-month dispensing reduces the need for monthly clinic visits and, since 2016, the World Health Organization has recommended this for stable patients as part of differentiated service delivery. The aim of differentiated care is for people with less complex medical needs to receive more care in the community, freeing up medical resources for people starting treatment and those with more complex needs.
At the virtual conference, a poster outlined the experience of 14 African countries, based on information provided by experts in their ministries of health in April and May.
Within weeks of the first reported cases of COVID-19 in Africa, all the countries had made changes to their differentiated care programmes and ten countries changed policies so that more people were eligibile for multi-month dispensing. Some patients have moved from three-monthly to six-monthly refills. However, South Africa suspended its plans to expand multi-month dispensing due to fears of ART stock outs.
Other models of differentiated care have had to be scaled back, with adherence clubs and community ART groups (which involve groups of patients meeting regularly and receiving their medication at the same time) being modified or suspended in 12 countries.
In a separate piece of research, adherence clubs in Khayelitsha, Cape Town, were randomised to either two or five meetings a year. A total of 2150 patients in 88 adherence clubs were included, with outcomes assessed after 24 months. There were no significant differences in terms of retention in care or viral suppression. Patients reported finding the less frequent meetings more convenient and flexible.

Dolutegravir in pregnant women living with HIV
Neural tube defects are severe birth defects of the brain, spine or spinal cord. In May 2018, the Tsepamo study, an ongoing birth outcomes surveillance programme in Botswana, unexpectedly identified an association between dolutegravir use at conception and neural tube defects. Findings from other studies were inconclusive, highlighting the overall lack of safety data on antiretrovirals in pregnancy.
Dr Rebecca Zash presented updated data from Tsepamo: an additional 39,200 births have been recorded since the last update, bringing the total to 158,244 deliveries. The prevalence of neural tube defects among infants born to women on dolutegravir at the time of conception appears to be stabilising at approximately 2 in 1000 (0.2%) deliveries compared to other antiretrovirals at 1 in 1000 (0.1%).
“Women are not a niche population; to maintain gender equity requires pregnancy safety data,” Zash commented. “If we don’t include women in trials, we are not protecting women.”
The conference also heard the results of a meta-analysis of five recent randomised clinical trials in sub-Saharan Africa in which pregnant women took a dolutegravir-based or efavirenz-based regimen. The safety profiles were similar, but there was greater viral suppression in women taking dolutegravir.
Depo Provera may worsen tenofovir-related bone loss
Previous research has shown that people living with HIV experience accelerated loss of bone density and are at greater risk for osteopenia (low bone mineral density) and osteoporosis (more severe loss of bone density that can lead to fractures). TDF is highly effective and considered generally safe, but it can cause loss of bone density in susceptible individuals.
The widely used injectable contraceptive depot medroxyprogesterone acetate (DMPA), also known as Depo Provera, leads to lowered oestrogen levels that can also cause loss of bone density.
Researchers in Uganda investigated the combined effect of TDF and Depo Provera on bone mineral density, recruiting women aged 18 to 35 from health facilities in Kampala.
They compared 159 HIV-positive women starting first-line ART containing TDF who were using Depo Provera, 106 HIV-positive women starting TDF-containing ART who were not using hormonal contraception and 69 HIV-negative women.
Bone mineral density was assessed every six months for two years. At the start of the study, the HIV-positive women's bone density was not significantly different from that of the HIV-negative women. Over time, however, the rate of bone loss was greater in the HIV-positive women, with the greatest loss seen among those using Depo Provera.
The researchers suggest that alternative contraceptive methods and newer antiretroviral regimens are needed to mitigate bone loss among young women with HIV.

Many people in African PrEP programmes stop and restart PrEP
Over 47,000 people were enrolled in the three projects in Kenya, Tanzania and Lesotho. Participants were mostly young people and members of key populations such as female sex workers and men who have sex with men.
The analysis assumed that if people returned for a PrEP prescription more than 14 days after daily PrEP would have run out, then they must be using it episodically. Their new prescription was counted as a PrEP restart.
Fifty-five per cent of people initially attending for PrEP never returned. Of those who did, almost half returned promptly for PrEP prescriptions and were therefore presumably using PrEP daily. This left 53% who had at least one PrEP ‘restart’.
Of those restarting, 66% stopped and restarted only once, 20% stopped twice, and 14% stopped three or more times. The biggest independent predictor of time off PrEP was younger age.
“Cycling on and off PrEP repeatedly is common," Jane Mutegi of Jhpiego told the conference. "Providers should anticipate episodic use and not assume indefinite, continuous use is the only effective use, or the best dosing option for all.”
A separate analysis surveyed 193 young women in Lesotho about their HIV risk perception and their reasons for stopping and sometimes restarting PrEP. Paradoxically, women who discontinued PrEP were 3.4 times more likely to perceive themselves to be vulnerable to HIV. Why did they stop? The most common reason was a bad encounter with a health provider (27%), while 21% said they’d stopped due to side effects. “I don’t think I’m at risk of HIV” was only cited by 14%.
aidsmapLIVE: AIDS 2020 special

On Thursday 9 July, we broadcast an aidsmapLIVE AIDS 2020 special. Joining NAM’s Susan Cole to discuss the biggest stories from the conference were: Dr Laura Waters, Chair of the British HIV Association; Professor Linda-Gail Bekker, Deputy Director of the Desmond Tutu HIV Centre; AIDS 2020 plenary speaker Lucy Wanjiku Njenga; and NAM aidsmap’s Gus Cairns.
If you missed the live show, you can watch it on aidsmap.com and our social media pages.
Scientific analysis from Clinical Care Options

Clinical Care Options is the official provider of online scientific analysis for the 2020 International AIDS Conference through capsule summaries, downloadable slides, rapid expert webinars, and ClinicalThought commentaries.
