The novel HIV attachment inhibitor fostemsavir continues to maintain viral suppression in more than half of people with extensive prior treatment experience and highly resistant virus, according to a report at the 10th International AIDS Society Conference on HIV Science (IAS 2019) last week in Mexico City. Study participants, many of whom had advanced immune suppression at baseline, experienced substantial gains in CD4 cells.
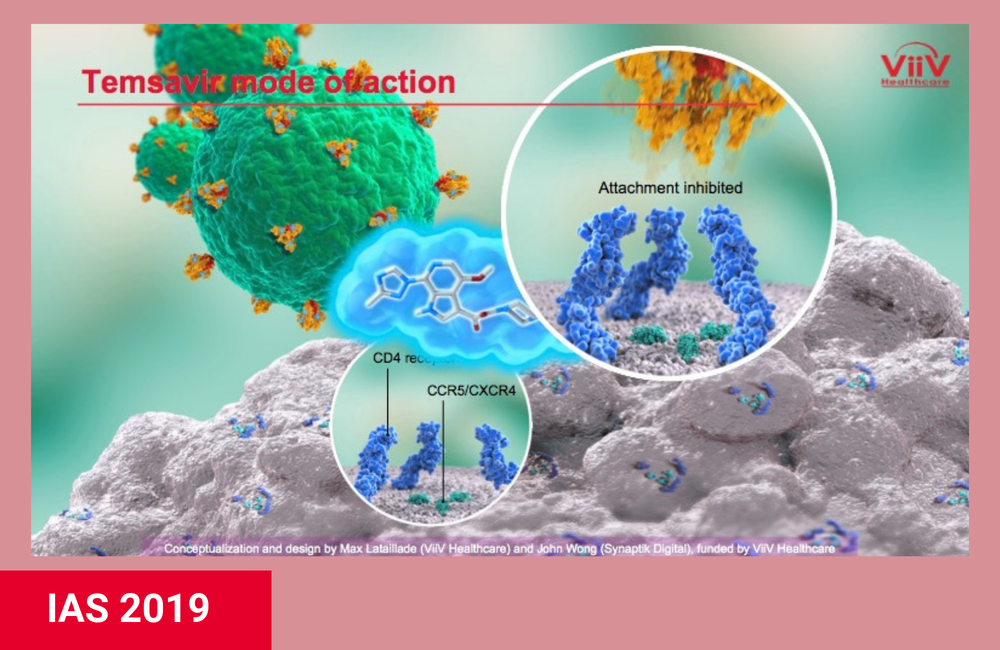
Fostemsavir is the first oral antiretroviral that prevents HIV from attaching to host cells. Its active form, called temsavir, binds to the gp120 portion of the HIV envelope protein that makes up the spikes on the surface of the virus. This prevents the virus from attaching to the CD4 receptor on T cells and other immune cells, which it uses to gain entry into the cells. (The injectable drug enfuvirtide [Fuzeon] binds to the gp41 portion of the HIV envelope protein.)
Dr Max Lataillade of ViiV Healthcare presented the latest findings from the phase 3 BRIGHTE study, an ongoing trial evaluating fostemsavir in heavily treatment-experienced people with multidrug-resistant HIV who were unable to construct an effective antiretroviral regimen using existing drugs.
The randomised portion of the study included 272 participants on a failing regimen with unsuppressed virus (at least 400 copies/ml). They were assigned to receive either 600mg twice-daily fostemsavir or a placebo along with their failing regimen for eight days. After that, everyone received open-label fostemsavir plus an optimised background regimen containing the most active drugs available to them. All participants had to have one or two remaining antiretroviral drug classes with at least one fully active approved agent; dolutegravir was still fully active for about 65%.
In this cohort, about three-quarters were men, most were white and the median age was 48 years; 60% were over age 50. Most had a viral load in the 1000 to 100,000 copies/ml range, but 29% had over 100,000 copies/ml. The median CD4 count was 99 cells/mm3, with 72% having fewer than 200 cells/mm3 and 26% having fewer than 20 cells/mm3. Most had a history of AIDS.
In addition, 99 individuals who had no remaining fully active approved drugs were included in a non-randomised compassionate use cohort. This group could use other investigational agents in their optimised regimen, such as ibalizumab (which has since been approved in the US as Trogarzo and recently received a positive opinion from the European Medicines Agency). This cohort was demographically similar but had more advanced immune suppression, with a median CD4 count of 40 cells/mm3.
As reported at the 2017 European AIDS Conference, fostemsavir used as functional monotherapy reduced viral load more than placebo during the first eight days. HIV RNA decreased by a median 1.0 log10 in participants with baseline viral load above 1000 copies/ml, Lataillade said in Mexico City, although he previously reported the drop as 0.8 log10.
Lataillade presented 48-week findings at last year's International Congress on Drug Therapy in HIV Infection in Glasgow. At that point, 54% of participants randomised to receive fostemsavir and 38% of those in the non-randomised cohort achieved viral suppression (< 40 copies/ml).
By week 96, the proportion of randomised patients with viral suppression rose to 60% in an intention-to-treat analysis. People who changed their optimised background regimen for efficacy reasons were counted among the 30% who experienced virologic failure. The other 10% had no virologic data due to treatment discontinuation or death. In an observed or as-treated analysis, the viral suppression rate was higher, with 79% having less than 40 copies/ml and 88% having less than 400 copies/ml.
In the non-randomised cohort, the 96-week viral suppression rate remained stable at 37% by intention-to-treat analysis. Here, 43% experienced virologic failure and 19% had no virologic data. In the observed analysis, 59% had HIV RNA less than 40 copies/ml and 68% had less than 400 copies/ml.
Viral load reduction was accompanied by improved CD4 counts. The mean gain at 96 weeks in the randomised cohort was 205 cells/mm3 – an increase of 66 cells/mm3 from week 48. The greatest improvement (240 cells/mm3) was seen in those with less than 20 cells/mm3 at baseline. Among the 71 people who started with less than 50 cells/mm3 at baseline, 56% had at least 200 cells/mm3 at week 96, the threshold for an AIDS diagnosis and increased risk of opportunistic infections. The mean CD4-to-CD8 cell ratio also rose. In the non-randomised cohort, the mean CD4 increase was 119 cells/mm3.
Treatment continued to be safe and well tolerated. Looking at both cohorts together, 21% experienced drug-related moderate to severe (grade 2-4) side-effects, 3% had drug-related serious adverse events and 7% stopped treatment due to adverse events. The most common adverse events were nausea, diarrhoea and headache. In addition, there were 38 (10%) AIDS-defining events and 29 (8%) deaths in this highly immunocompromised population; all but six of the deaths were in people with less than 50 cells/mm3.
"Virologic response continued to improve over time despite continued attrition in this difficult-to-treat population," the study investigators concluded. "BRIGHTE results support continued development of fostemsavir as an important treatment option for heavily treatment-experienced people living with multidrug-resistant HIV-1."
These findings show that fostemsavir "can be life-changing for people living with HIV and a low CD4 count," Lataillade said.
In response to questions from advocates, he acknowledged that the fostemsavir early access programme (EAP) had been limited and that filing for US Food and Drug Administration (FDA) approval had been delayed by manufacturing issues. However, two new manufacturing facilities were recently completed and ViiV expects to be able to reopen the EAP and is hoping to file for FDA approval by end of this year, according to Lataillade.
Lataillade JM et al. Week 96 safety and efficacy of the novel HIV-1 attachment inhibitor prodrug fostemsavir in heavily treatment-experienced participants infected with multi-drug resistant HIV-1 (BRIGHTE study). 10th International AIDS Society Conference on HIV Science, Mexico City, abstract MOAB0102, 2019.
View the abstract on the conference website.
Update: Following the conference presentation, this study was published in a peer-reviewed journal:
Lataillade JM et al. Safety and efficacy of the HIV-1 attachment inhibitor prodrug fostemsavir in heavily treatment-experienced individuals: week 96 results of the phase 3 BRIGHTE study. The Lancet HIV, 7:e740-e751, November 2020.
