Pronounced racial HIV disparities persist in the US

Professor James Hildreth made a strong case for attending to social determinants of health in order to address pronounced racial disparities in both the HIV and COVID-19 pandemics in the US, during a plenary session at the virtual Conference on Retroviruses and Opportunistic Infections (CROI 2021).
Poor access to health information, delayed medical care, poorly managed chronic conditions, mass incarceration and economic instability are some of the factors which act as social determinants of health, he said.
New data presented at the conference by Dr Jun Li from the Centers for Disease Control and Prevention (CDC) indicated that while racial differences have decreased from 2012 to 2018, Black people living with HIV are still significantly less likely than White counterparts to receive antiretroviral therapy within a month of entering care.
The Deep South has the highest rates of new HIV diagnoses, the largest number of people living with HIV and the lowest rates of viral suppression in the US. A separate study from Alabama, Louisiana and Mississippi indicated that while overall viral suppression trends are improving, young Black men continue to take longer to achieve viral suppression than other groups.
A third study demonstrated how increased funding in certain jurisdictions provided an opportunity to correct some of these disparities. The THRIVE demonstration project supported areas with high numbers of Black or Latino men living with HIV to develop comprehensive community-based HIV prevention and care services, including activities designed to support PrEP implementation.
There was an estimated 4.2% reduction in new HIV diagnoses among Black men who have sex with men from 2014 to 2018 in THRIVE-funded jurisdictions, whereas comparable areas not receiving funding saw a consistent number of new diagnoses. For Latino men who have sex with men, there was a 1.7% increase in those cities not receiving funding compared to a 2.7% reduction in those that did.
African study supports second-line dolutegravir and tenofovir recycling
Dolutegravir or darunavir/ritonavir are equally effective in second-line treatment, even in the presence of high-level resistance to nucleoside reverse transcriptase inhibitors (NRTIs) included in the regimen, a large randomised study carried out in three African countries has found. Furthermore, tenofovir and lamivudine can be recycled in second-line treatment, even in people with drug-resistance mutations, simplifying the process of switching treatment, the study presented at CROI 2021 by Professor Nicholas Paton showed.
The results are relevant to current World Health Organization (WHO) guidance, which recommends that people who experience viral rebound on a first-line regimen containing efavirenz or a boosted protease inhibitor can switch to dolutegravir and two NRTIs. However, WHO recommends including a new NRTI in the regimen and as most people on first-line treatment now take tenofovir, this means switching to zidovudine, which is less well tolerated.
The study recruited 464 people living with HIV at seven sites in Kenya, Uganda and Zimbabwe. They had viral loads above 1000 copies/ml on a failing NNRTI-based first-line regimen containing tenofovir and lamivudine. They were randomised to switch to either dolutegravir or darunavir/ritonavir (800/100mg), and then in a second randomisation, to either retain tenofovir or switch to zidovudine.
Twenty-seven per cent had viral loads above 100,000 copies/ml, indicating advanced failure of HIV treatment and there were high levels of drug resistance (92% had resistance to lamivudine, 58.5% to tenofovir and 18% to zidovudine).
The lack of active NRTIs in the regimen did not lead to differences in response between dolutegravir and darunavir: 90.2% of people taking dolutegravir and 91.7% of people taking darunavir had a viral load below 400 copies/ml at week 48. There was no significant difference in virological outcome according to NRTI randomisation; 92.3% in the tenofovir group and 89.6% in the zidovudine group had a viral load below 400 copies/ml at week 48.
While the results support the WHO recommendation of a switch to dolutegravir in second-line treatment, they suggest a revision to WHO guidance on NRTIs.
The study findings suggest that implementing resistance testing in resource-limited settings within current frameworks for changing to second-line treatment is unlikely to improve viral suppression and that a continued emphasis on viral-load testing and intensified adherence remains critical in managing first-line treatment failure.
Growing old with HIV in the US
The numbers of Americans living with HIV who are over 65 is projected to grow rapidly over the next decade and will result in large numbers who have multiple co-morbidities in addition to HIV, Dr Parastu Kasaie of Johns Hopkins University told CROI 2021.
The figures come from mathematical modelling, for which key inputs included CDC surveillance data and the incidence of co-morbidities in the North American AIDS Cohort Collaboration on Research and Design (NA-ACCORD), which includes over 130,000 people living with HIV.
The model projects that, by 2030, over 25% of people taking HIV treatment will be over the age of 65. Half will be over the age of 53.
By 2030, 36% of people taking antiretroviral therapy are expected to have multimorbidity – in other words, at least two physical co-morbidities in addition to HIV. Among people over the age of 70, the prevalence of multimorbidity is expected to increase to 69% by 2030.
The model predicts substantial increases in the prevalence of anxiety (from 36% to 48%), chronic kidney disease (from 16% to 26%), diabetes (15% to 24%) and myocardial infarction (3% to 9%). More encouragingly, depression, raised lipids, hypertension, cancer and end-stage liver disease are projected to only increase slightly or to decrease.
Reformulated islatravir implant should provide PrEP for over a year
A new formulation of the drug islatravir in the form of a small removable implant should provide enough of the drug to act as PrEP or form part of combination antiretroviral therapy for well over a year, Dr Randolph Matthews of Merck told CROI 2021.
This second phase I drug-level and safety study tested implants containing 48mg, 52mg and 56mg of islatravir in 24 volunteers, alongside a placebo implant in 12 volunteers. The volunteers kept the implants in for three months, after which they were removed.
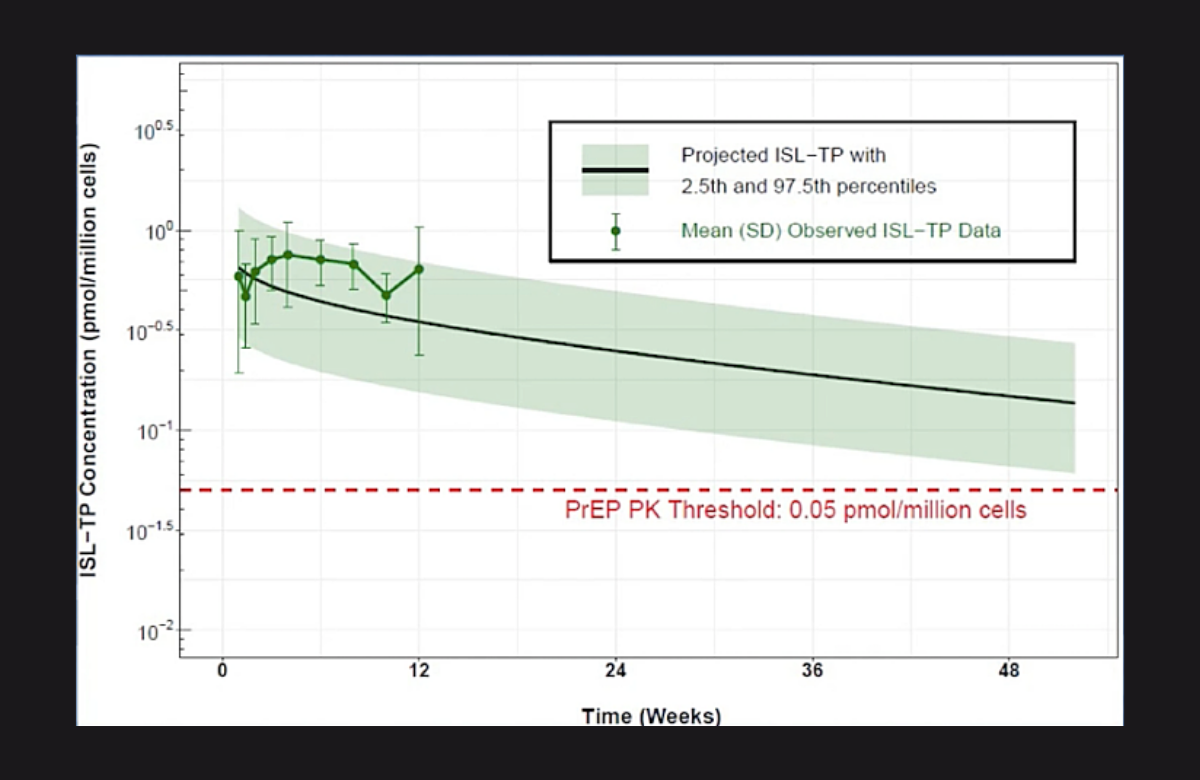
Separate studies had shown that a level of 0.05 picomols of drug per million circulating T-lymphocyte immune cells (0.05 pmols/106 cells) should be more than adequate for preventing HIV infection.
After removal, the intracellular half-life of the remaining islatravir in the body (the rate at which it tailed off) was 198 hours, meaning that after 8.5 days there was half as much drug left as when the implant was removed. In the two smaller doses, drug concentrations persisted above the 0.05 pmols/106 cells level for no more than two weeks but in the largest dose they persisted for one to two months.
Projections from this three-month study predict that after a year, the average drug level in people using it would still be 3-4 times above the 0.05 pmols/106 cells level. A phase II study is planned, which will establish whether real-life drug levels are the same as forecast.
Alendronate can prevent bone loss in people starting HIV treatment
A short course of alendronate around the time of starting tenofovir-based antiretroviral therapy can help prevent bone loss, according to a study presented at CROI 2021.
Bone loss – osteopenia and its more severe form, osteoporosis – is a common metabolic complication among people with HIV, especially older individuals. Bone loss is worsened by chronic inflammation and is associated with certain antiretrovirals, in particular tenofovir disoproxil fumarate (TDF).
Dr Tara McGinty of University College Dublin and colleagues designed the APART study to evaluate whether short-term use of a generic formulation of the oral bisphosphonate alendronate (branded as Fosamax) could prevent bone loss in people starting HIV treatment.
The study enrolled 50 people with HIV who were starting treatment for the first time, with antiretroviral regimens containing TDF, emtricitabine and a third drug, usually an integrase inhibitor. The participants were randomly assigned to receive 70mg oral alendronate or a placebo, once a week, beginning two weeks prior to starting HIV treatment up to week 14.
Most participants (86%) were men, and the median age was 35 years (range 32 to 40 years). This is of note particularly as older people – especially post-menopausal women – are at high risk for bone loss.
At 14 weeks, a difference between the two groups was already apparent. People assigned to alendronate had a median +1.88% increase in total hip bone mineral density (BMD) compared with a -0.65% decrease in the placebo group. BMD declined slowly and steadily in both groups, ending up with a +0.50% increase in the alendronate group versus a -2.70% decrease in the placebo group at week 50.
The impact on BMD at the lumbar spine was less durable. At 24 weeks, there was a +0.50% increase in the alendronate group versus a -2.50% decrease in the placebo group. But by week 50, this fell to a -1.40% decrease and a -3.70% decrease in the two groups, respectively, and the difference was no longer statistically significant.
Given that the drug is inexpensive, easily available and well tolerated, the researchers concluded that these data support the use of short-course generic alendronate to preserve BMD in people starting HIV treatment, including in resource-limited settings.
Large numbers on Descovy PrEP in the US
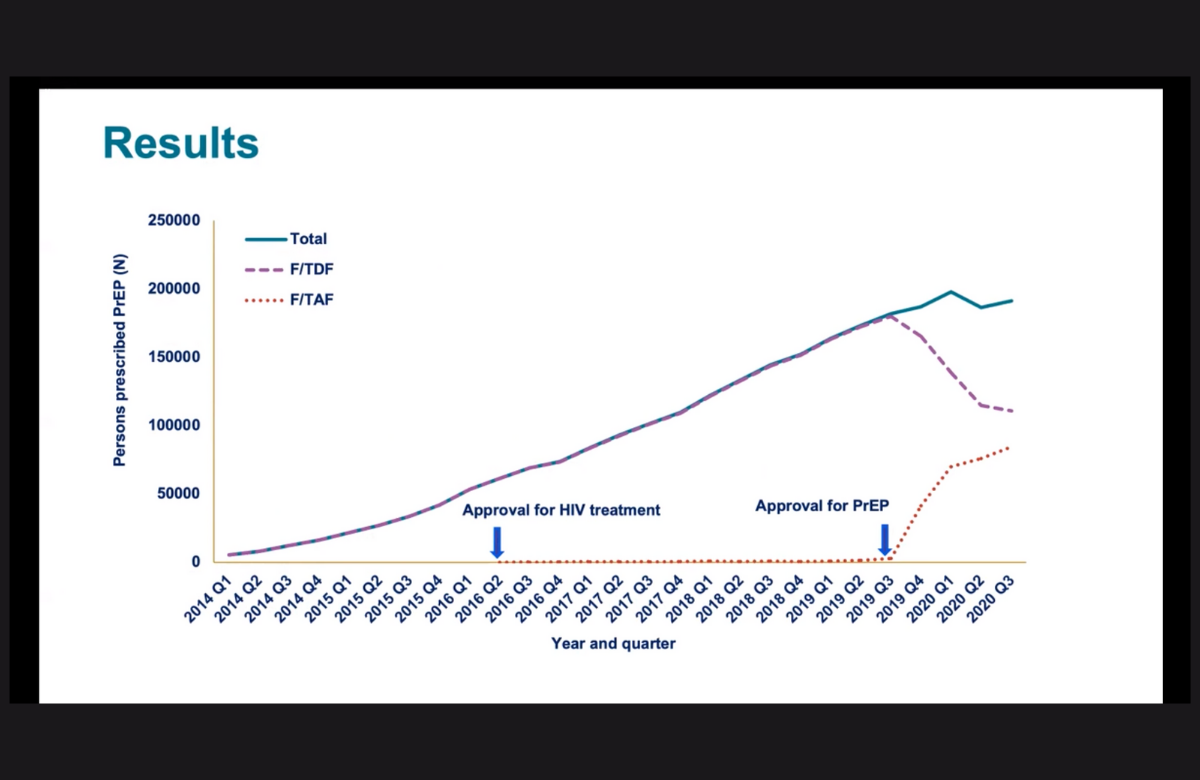
In October 2019, a newer formulation of PrEP (regular medication to prevent HIV infection), made up of emtricitabine and tenofovir alafenamide (TAF) and marketed under the brand name Descovy, was approved in the US. In the first year, 29% of people already taking PrEP switched to the newer formulation and 36% of people starting PrEP for the first time started on it. Dr Karen Hoover of the CDC presented these data, based on a large pharmacy database, to CROI 2021.
TAF-based PrEP has been a marketing success in the US, but it has not yet been approved by other regulatory agencies due to a lack of data in cisgender women. Moreover, given that emtricitabine/TDF is now widely available as a low-cost generic, health systems which are attentive to questions of cost effectiveness are likely to see Descovy as a niche option for individuals who have kidney problems or other specific concerns, rather than the default.
