The same messenger RNA (mRNA) approach used for the highly effective Pfizer-BioNTech and Moderna COVID-19 vaccines also shows promise for protection against HIV, according to a presentation this week at the virtual Conference on Retroviruses and Opportunistic Infections.
Despite more than three decades of research, scientists have had little success developing vaccines to prevent HIV. To date, only one vaccine regimen – a canarypox vector primer followed by a gp120 protein booster – has demonstrated partial protection in human studies; however, it was not effective in a recent large trial.
Two other large studies, Mosaico and Imbokodo, are currently testing an approach that uses an adenovirus primer followed by a booster that contains a 'mosaic' of proteins from multiple HIV strains. This is similar to the approach used for the Johnson and Johnson, Oxford-AstraZeneca and Russian Sputnik COVID-19 vaccines.
Dr Peng Zhang of the US National Institute of Allergy and Infectious Diseases (NIAID) and colleagues, in collaboration with Moderna, are taking a different approach that uses lipid nanoparticles, or fat bubbles, to deliver small lengths of a nucleic acid that delivers instructions for making proteins. mRNAs are found throughout the cell and act as messengers, hence the name.
They do not form part of the genome, or basic blueprint for life, that resides in the cell’s nucleus in the form of a similar but more stable molecule, DNA.
The investigators designed a vaccine regimen that introduces mRNA into a cell and instructs it to make the outer, envelope proteins of three different subtypes of HIV, plus the structural gag protein from SIV, HIV's simian cousin. The cells then assemble these proteins to make virus-like particles that trigger an immune response. These particles are not viruses because they have no genetic material of their own and cannot reproduce – but they look like viruses to the immune system.
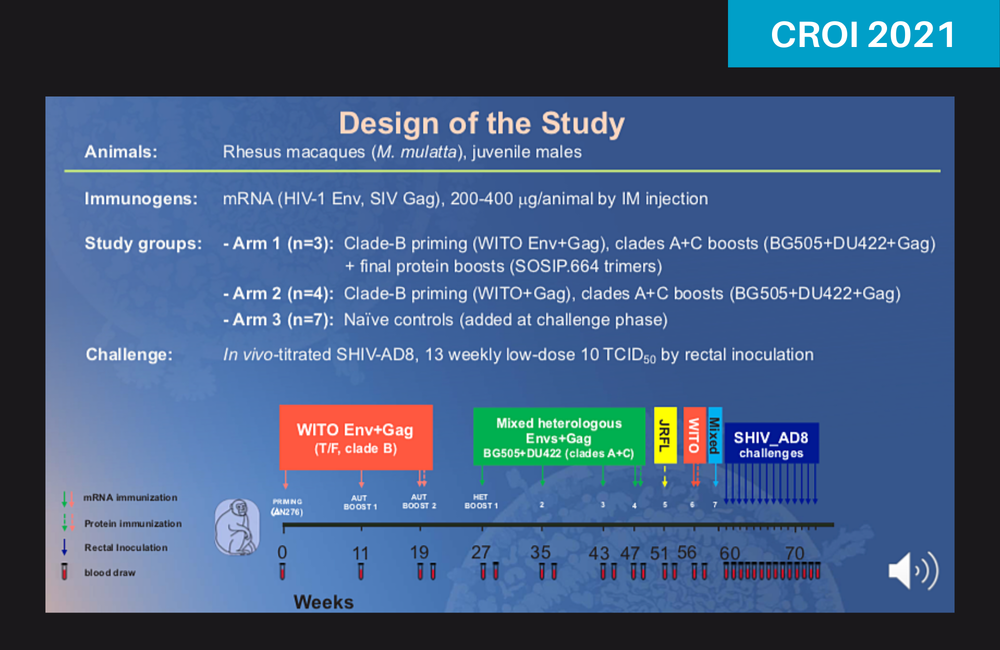
In this study, seven male macaque monkeys received one of two vaccine regimens while another seven received placebo injections.
All of the monkeys first received three injections over 20 weeks of a vaccine containing mRNA for HIV subtype B envelope proteins, the predominant type in Europe and North America. They then received four injections over 20 weeks of a second vaccine mixing mRNA from HIV subtypes A and C, which are predominant in eastern and southern Africa.
In addition, three monkeys also received final boosters containing stabilised HIV spike proteins known as SOSIP trimers, which maintain the protein in an open shape that exposes a conserved region that is more easily recognised by broadly neutralising antibodies. (The general approach, used in an earlier study by the same team presented at the International AIDS Conference last summer, is described in more detail here.)
After receiving the full vaccine regimens, the monkeys were challenged with 13 weekly rectal exposures to a tier 2 SHIV, an engineered HIV/SIV hybrid virus. Tier 1 viruses have a more open envelope trimer shape and are more easily recognised by neutralising antibodies, while tier 2 and 3 viruses have a more closed shape and have moderate or low sensitivity to these antibodies; most circulating HIV strains are classified as tier 2.
The vaccinated monkeys rapidly produced high levels of antibodies that bound to the protein trimer as well as neutralising antibodies against subtype B HIV proteins – the ones in the first set of vaccines. Starting at around 49 weeks, they produced lower levels of cross-reactive neutralising antibodies against the subtype A and C proteins. By the end of the immunisation regimen, the monkeys had produced broad-spectrum tier 2 neutralising antibodies against a range of viral strains.
All seven animals in the placebo group became infected within about a month and a half, starting after the second exposure.
All vaccinated monkeys remained free of SHIV for about the first month, but then started to become infected; a few, however, remained uninfected at two months, after all the exposures.
This worked out to be an 85% reduction in the risk of infection overall, a 76% reduction for the monkeys that received the mRNA vaccines alone, without a booster, and an 88% reduction for those that got the mRNA vaccines plus the trimer protein boosters. Protection was correlated with the presence of antibodies to the CD4 binding site, which HIV and SIV use to enter cells.
At a press briefing, Dr Zhang said: "The mRNA platform represents a very promising approach for the development of an HIV vaccine in the future."
Putting the findings in simpler terms, the mRNA vaccine, combined with the stabilised booster protein, was able to "open up the CD4 binding site," in the words of Dr Richard Koup of NIAID's Vaccine Research Center, who moderated the briefing.
Ultimately, the monkeys "developed reasonable neutralising activity and were then protected partially from infection," he added. "This provides a new avenue, potentially, for moving this into human vaccination."
Zhang P et al. An Env-Gag mRNA vaccine protects macaques from heterologous tier-2 SHIV infection. Conference on Retroviruses and Opportunistic Infections, abstract 86LB, 2021.
Download the abstract eBook from the conference website.
Update: Following the conference presentation, this study was published in a peer-reviewed journal:
Zhang P et al. A multiclade env–gag VLP mRNA vaccine elicits tier-2 HIV-1-neutralizing antibodies and reduces the risk of heterologous SHIV infection in macaques. Nature Medicine, 27: 2234-2245, December 2018.