Eighteen months ago, aidsmap.com reported on a study that suggested a mechanism behind some people’s ability to control their HIV without drugs. A new study, published in the scientific journal Cell, used sophisticated probes to uncover every bit of HIV genetic material hidden in the cells of six patients. It goes a long way towards finding out how this happens naturally – which is a first step in finding out how to ‘Block and Lock’ ongoing HIV infection permanently.
Dr Mathias Lichterfeld, a member of the research team, commented in Science magazine that their findings raise the possibility of “a peaceful coexistence between HIV and humans.”
Background – reservoirs and controllers
During HIV infection, HIV doesn’t simply subvert cells into becoming factories for new virus, in the way that most viruses do. As a retrovirus, it is able to splice its genes into our human DNA in the nucleus of our cells, thereby becoming ‘part of us’ in a much more intimate way.
Some of these cells are the long-lived Central Memory T-lymphocytes – the ones whose job it is to remember old infections, or the things that look like old infections we call vaccines.
Some of these cells, in people who are on antiretroviral therapy (ART) and therefore don’t have obvious, progressive disease, harbour hidden instructions for making new HIV that are invisible to the immune system. They truly are sleeper cells, harbouring a so-called reservoir of inaccessible HIV.
In most people, as soon as ART is stopped, these reservoir cells start making viruses again. But in a few people, they don’t. Why?
Some people – about one in 200 with HIV – are the so-called ‘elite controllers’, who remain virally undetectable without ever taking antiretroviral therapy (ART) at all.
In two people – a Californian called Loreen Willenberg and the Esperanza Patient, named after her Argentinian home town – this process seems to have reached its ultimate point; both seem to have essentially cured themselves of HIV without medical intervention.
A larger but still undetermined number of people are the so-called ‘post-treatment controllers’. These are people with progressive HIV infections and detectable viral loads who start ART but who then, by choice or accident, stop their meds – and remain undetectable for months or years thereafter.
There may be many more potential post-treatment controllers than we know of because, understandably, most people on long-term ART don’t stop it. The original French study investigating them suggested it could be as many as one in 14 people on ART. In one case of, coincidentally, another Argentinian woman reported last year, she had initially had severe HIV disease and had been on ART for a decade. She stopped it due to side effects and, 12 years later, has minimal signs of HIV and has not had to restart it.
The study we reported on 18 months ago was important because, for the first time, it suggested an underlying cause for the viral control seen in controllers – and thereby suggested routes towards a cure.
It looked for HIV gene sequences hidden in the DNA inside the immune cells of 64 elite controllers. It found fewer lengths of DNA in them than in other people with HIV, as expected. But it found a higher proportion of DNA that was intact and capable of giving rise to new viruses.
But what was crucial was where that DNA was located. It seemed to be parked in so-called ‘gene deserts’ – parts of the genome that consist of junk or structural DNA but which aren’t active genes. This DNA could never be woken up.
The researchers suggested that in elite controllers, from the start, or in post-treatment controllers, during years on ART, the immune system spotted subtle, intracellular signs of viral replication – most notably, bits of viral RNA that are ‘read off’ the DNA and message the cellular machinery to start making new viruses. This way, the cells containing the most transcriptionally active viral DNA are selectively killed by the immune system’s CD8 cells – just leaving the permanently ‘blocked and locked’ cells.
The new study
The research team is led by Drs (and spouses) Xu Yu and Mathias Lichterfeld at the Ragon Institute, a cutting-edge collaboration between Harvard University, the Massachusetts Institute of Technology, and Massachusetts General Hospital. In the 2019 report we noted that Ragon “is now contacting people who have been on stable antiretroviral therapy (ART) for more than 20 years to find out if they too, have managed to exile their virus to a genetic version of Siberia.”
The new study tells us about six such people who are on long-term ART. The Ragon team used the latest technology to conduct a deep genetic probe into the HIV DNA hidden within the T-lymphocyte immune cells of these six volunteers.
The study participants were aged 54 to 74. One was a woman and one had no gender recorded: the others were men. All had been on ART by at least nine years at the date of their last cell sample. They were chosen because they had had cell samples taken as early as 2001 that were suitable for deep sequencing. Three had had samples taken before starting on ART or, in one case, during an interruption. Four had samples taken a year after starting ART, and all six had a sample taken between nine and 15 years after starting ART, three very recently.
The sophisticated “Parallel RNA, Integration and Proviral Sequencing” (PRIP-seq) assay they used took millions of free-floating lymphocyte cells in the patients’ blood (PBMCs) and diluted them down till they had several thousand cells in each sample. As within the cellular reservoir only one in ten thousand to one in a million cells or even fewer contains HIV DNA, this meant they were able to detect individual sequences of HIV DNA within single cells.
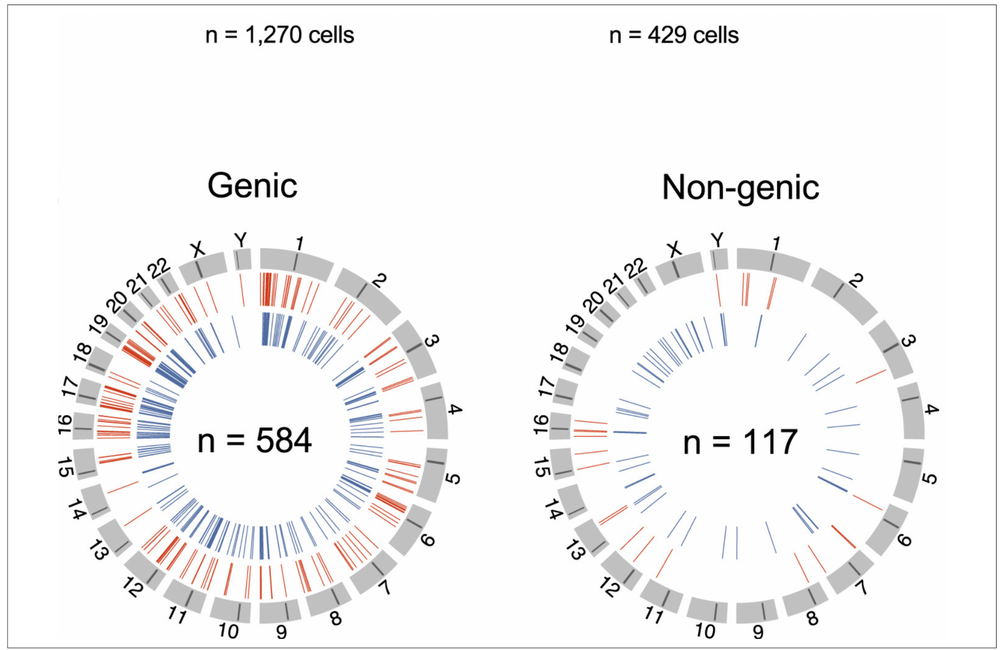
They were able to find individual lengths of HIV DNA from 1270 specific cells. Most of these ‘proviruses’ were in fact defective and incapable of giving rise to whole viruses; they found 147 lengths of complete, intact HIV genome in the six patients – extraordinarily few compared to the millions of cells sampled.
They found 429 cells that, at one or more time points, were actively producing measurable amounts of HIV RNA, and were therefore actively transcribing HIV ‘instructions’, whether or not those instructions were complete. In some circumstances, even if their RNA instructions are incomplete, viruses can reassemble.
The PRIP-seq probe didn’t just look for viral DNA. It can also see exactly where, within the human genome, each length of HIV is inserted – and therefore whether it is near an active site where host genes are often ‘awake’ and producing their own RNA messages, or whether it is marooned in one of the ‘gene deserts’.
The scientists found out that HIV could integrate itself pretty much anywhere in the genome. They found proviruses in every one of the 23 pairs of chromosomes in each of the patients, in active sites and in locked sites. it wasn’t just where the HIV DNA was linearly within the human DNA that mattered, either. Proximity to active genes was a matter of three dimensions, because human DNA contains about three billion individual bases (‘letters’) in its chain and has to be folded up intricately. Functional proximity could therefore happen not just within each chromosome, but even between chromosomes.
The researchers found 701 cells where, in initial samples, they could establish the exact integration site and found that in 117 cells, the proviral DNA was in an area that did not contain functional genes. Even in these, viral RNA was detectable in some 22 cells, but it was very unlikely to be functional.
The strongest determinant of an HIV-infected cell producing HIV RNA was that its HIV DNA was near an active human gene, or an area of human DNA that switched on genes. Conversely, the second-strongest determinant of it not producing RNA was that it was near an area that was locked up into an inactive form (by a process called methylation) or near an area of human DNA that made that happen.
But the strongest predictor of cells not producing HIV RNA was the length of time the study subjects had been on ART – thus confirming the researcher’s hypothesis that functional HIV DNA capable of being transcribed becomes rarer over time.
There were only three patients that had several PBMC samples taken over time and in whom, therefore, longitudinal proviral DNA decay could be measured. The proportion of cells producing viable HIV RNA fell fivefold in two patients whose cells were sampled nine years after the first assay, and to virtually nothing in the one subject sampled 12 years later. A fourth patient only had one cell sample taken, 15 years after they started ART, and this patient also had no evidence of RNA production.
So at the individual level, they found evidence of the ‘blocking and locking’ of viral DNA in four out of six patients. They believe that this process happens because, even under ART, the immune system can detect the very low levels of HIV RNA that cells are producing and selectively kill the most active ones, so only the cells with blocked viral DNA are left.
They point out that ‘kick and kill’ cure strategies that have relied on activating the entire cellular reservoir have produced largely disappointing results because all they have done is to count whether they have produced an overall reduction in central memory T-cells containing viral DNA – and they haven’t.
If, however, activation does something more subtle – a selective culling of the cells that produce the most RNA – then such strategies could end up with a reservoir of cells that still contain HIV DNA, but none that can start producing viral components. Even when cells from the four patients were stimulated in the lab dish with powerful gene-activating chemicals, they did not produce any HIV RNA.
This left two people in whom the story was rather different, with no decline in levels of viral RNA being produced.
"This is a first step in finding out how to ‘Block and Lock’ ongoing HIV infection permanently."
It was found that in one, a lengthy piece of viral DNA was spliced into the middle of a human gene called KDM2A. This is an oncogene – a gene whose job is normally to stimulate cell division but which, it if is jammed on, can cause cancer. In the other, the viral DNA was not inside any human genes, but was near a length of DNA that is a powerful disrupter of chromosomal architecture, forcing the DNA to adopt a more open configuration that is permissive to genetic activation. This person had maintained a low but detectable viral load in the order of 20 copies/ml throughout their time on ART.
The question was: why hadn’t the same immune pressure eliminated these persistently active lengths of viral DNA?
The probable answer is that HIV infections don’t have to maintain themselves by activating, transcribing RNA and producing viral components. They can also do it without any production of RNA if they happen to be in an immune cell that divides and, in doing so, simply copies its DNA – including its viral DNA.
Because such so-called clonal expansion does not alert the immune system, the selective culling of active reservoir cells cannot happen and if the HIV DNA happens to be in a frequently-activated part of the human genome, it will stay there.
This implies that, as in other aspects of HIV infection, there is an evolutionary arms race between the immune system’s ability to remove cells productively infected with HIV and the same cells’ ability to copy themselves before elimination.
Finding the genetic signature that would indicate whether or not you are a post-treatment controller, and could risk stopping ART, requires sophisticated gene-sequencing tools, as in this study. So it’s not something ready for the clinic.
The next step for the Ragon team, however, is to screen patients on long-term ART for signs that all their proviral HIV DNA is sequestered into non-productive areas, and then take some off ART, using very careful medical monitoring so that ART can be restarted at the earliest sign of viral reactivation.
The report in Science says that one volunteer has already agreed to join such a strategy, and Yu and Lichterfeld are screening others.
Einkauf KB, Yu XG, Lichterfeld M et al. Parallel analysis of transcription, integration and sequence of single HIV-1 proviruses. Cell 185: P266-282.e15, 2022 (open access).