For the first time, the European AIDS Clinical Society (EACS) and the European Centre for Disease Prevention and Control (ECDC) have conducted an audit of one aspect of HIV clinical services, measuring provision in five disparate countries against one set of standards and comparing the degree to which different countries and clinics meet that standard. The screening, prevention and management of viral hepatitis in people with HIV was audited in Georgia, Germany, Poland, Romania and Spain.
The audit was a pilot project designed to demonstrate that such an exercise was possible. Even so, it represents a considerable leap forward in a long-standing project to develop common standards of HIV prevention and care across the entire World Health Organization (WHO) European Region, which stretches from western Europe to central Asia.
The EACS Standard of Care project started in Rome in 2014 and has now met four times. (Read our reports from Rome in 2014, Brussels in 2016 and Bucharest in 2019.)
A summary presentation of the first two meetings can be viewed here. They were largely concerned with how, and to what extent, EACS, as the regional association of physicians, could actively promote better and more equal standards of HIV testing and care throughout Europe, an area characterised by gross disparities in care.
Last year in Bucharest, considerable steps were taken towards developing a common, auditable European standard of care that could be fairly and realistically applied to clinics throughout Europe. The meeting resolved to develop a set of auditable standards against which clinics could be benchmarked.
The 2020 meeting, like most other conferences this year, has had to become a largely virtual one and has been split into an opening session, three workshops and a wrap-up meeting. The opening session took place last month. Initially the 2020 meeting had been planned for Tbilisi in Georgia and this session was partly live from that city, led by a team of physicians including Dr Tengiz Tsertsvadze, the country’s Director of Infectious Disease, AIDS and Immunology Research, with other EACS presenters joining online.
This meeting included a presentation of the pilot audit and also a summary of the effects of COVID-19 on HIV services across Europe – aidsmap.com will report the latter separately.
The three workshops will look further at the effects of COVID-19 (10 November), barriers to the delivery of PrEP (17 November) and co-morbidities and the ageing patient with HIV (27 January), with a wrap-up meeting in February.
At the opening meeting, former British HIV Association (BHIVA) Chair Professor Jane Anderson introduced the general concepts within which effective audits have to be framed. An audit is not a piece of research as such, she said. Audits are quality improvement activities. They deal not with best-case scenarios but with the possibilities for improvement that exist in current health care settings.
Audits have to provide “the right information, to the right people, at the right time”: ones that do not will not achieve their primary goal of service improvements.
They have to be aligned with existing and upcoming priorities and initiatives and be realistic about what improvements would cost now relative to how much money they might save later. Standards and priorities are not set in stone and might change. The COVID-19 epidemic was a prime example of this, with it necessitating the wider adoption of things like home testing and telemedicine. Some of these changes may be retained after COVID-19 and standards will change accordingly.
Setting up the audit: considerations and questions
Professor Chloe Orkin, current BHIVA Chair, said that audits of services could be powerful tools – catalysts for change on a bigger scale than that of individual clinics, or even countries. The UNAIDS 90-90-90 initiative was essentially a simple, auditable set of standards and had helped towards a global expansion of effective HIV health care.
The annual BHIVA audit, which served as part of the template for the EACS pilot, started in 2001 and had focused on a large variety of different clinical priorities: the first one audited CD4 count at diagnosis (a proxy for late testing) and the most recent audited provision of tests and services for ageing patients with HIV and co-morbidities. (See here for all of BHIVA's audit reports.)
Orkin talked about the practicalities involved in deciding on the subject and questions of an audit. Audits had to be relevant to existing guidelines, investigate systems that were already in place, and be feasible to answer (i.e. dealing with simple data that was collected in standard practice).
BHIVA’s practice is to send an audit questionnaire to every participating clinic and ask them to pull out approximately 40 patient records for a more detailed case note review. Overall, anonymised results are presented at BHIVA’s annual conference and a confidential report is sent to each clinic about its own results. A re-audit is carried out a year later to look at any resulting changes. Orkin showed examples from the 2018 audit on mental health services: a typical question from the case note review was 'Has the patient had a mental health assessment in the last 18 months?'
"The UNAIDS 90-90-90 initiative was essentially a simple, auditable set of standards and had helped towards a global expansion of effective HIV health care."
Dr Kamilla Laut of the Centre for Health, Immunity and Infections (CHIP) in Denmark presented the results of a scoping exercise designed to identify existing national and local standards of care, in order to identify the measures that would best be included in a European standard.
She said the global standards of care document that provided the best and simplest set of standards was the WHO’s Consolidated Strategic Information Guidelines for HIV in the Health Sector. This lays out the ten most important pieces of data to rate a country’s HIV response: the total number of people living with HIV and the number diagnosed with it; the annual number of new infections and AIDS-related deaths; care coverage and retention; HIV therapy coverage and viral suppression; the coverage of prevention services and, last but not least, the amount of domestic funding earmarked for HIV.
The scoping exercise asked for examples of national guidelines but also for useful background surveillance and policy documents.
EACS only received answers from seven countries: Denmark, Germany, Ireland, Romania, Spain, Switzerland and the UK, as well as documents from some pan-European agencies like ECDC. All countries’ guidelines covered antiretroviral therapy (ART) coverage, CD4 counts and viral load testing and suppression rates. Five out of the seven covered co-infections such as hepatitis; four covered co-morbidities such as kidney disease, and also covered pregnancy and post-exposure prophylaxis (PEP); three covered pre-exposure prophylaxis (PrEP), adherence and side effects. Three standards measured local performance against national benchmarks; two against local ones (e.g. in the same province); two more did not explicitly compare performances against any other clinics.
There was surprisingly little consensus as to which areas were the most important ones to cover. The ones that were mentioned most often were prevention, testing and diagnosis; complex HIV care; and outpatient care.
In detail, areas where three or more documents, both standards of care and policy documents, had included a particular standard, and where at least one of them gave a measurable indicator, included (this is not an exhaustive list):
Prevention
- Condom use in people living with HIV, the general public and priority populations.
- Percentage using PrEP in priority populations.
- Availability and number of sets of sterile injecting equipment for people who inject drugs.
Testing
- Percentage of people at risk of HIV with documented test offer.
- Percentage of specialist clinic attendees offered a test.
- Percentage of people testing HIV positive offered an appointment at a specialist HIV centre, and how soon.
Co-infection and co-morbidities
- Percentage screened for chronic and incident viral hepatitis.
- Percentage screened for latent and incident TB.
- Percentage screened for cardiovascular disease, hypertension, renal function, bone mineral density.
Limitations of the scoping exercise included that only seven countries, all but one from western Europe, answered it; that the existing standards of care mainly cover specialist clinical care, and fewer cover extra-clinical or referred services such as prevention or mental health; and that some standards of care documents may have been missed.
Nonetheless, Laut said, the scoping exercise showed that one common set of European standards of care should be feasible. Final topics would be decided by an expert group and audits conducted every one to two years. It would be a challenge to find the ten or so topics, and 2-3 key indicators per topic, that would be commonly applicable to all countries. In some cases it may be more realistic to measure local standards against other clinics or regional services in the same country.
The pilot audit: hepatitis A, B, C and D
Dr Ann Sullivan of London’s Chelsea and Westminster Hospital introduced the pilot audit that had been undertaken last year, as a joint project of EACS and ECDC. The aim of this was to demonstrate the feasibility and acceptability of an audit and the type of data that could be collected, though it did uncover some interestingly wide variations in provision within some countries, as well as between them.
The topic chosen was viral hepatitis screening, prevention and management among people with HIV. An audit data collection form was developed at the last standard of care meeting in Bucharest in 2019.
Five countries were chosen: Georgia, Germany, Poland, Romania and Spain. A total of 23 individual HIV clinics took part, with no country having fewer than four or more than six. In the audit, individual country and clinic results were ‘anonymised’ for public presentation, and were called countries A, B, C, D and E, with clinics individually numbered A1, etc.
The audit measured:
- Screening for hepatitis A, B and C viruses (HAV, HBV, HCV)
- Vaccination rates for hepatitis A and B in those not already immune
- Tenofovir therapy for hepatitis B vaccine non-responders
- Hepatitis delta (HDV) antibodies in people with chronic hepatitis B
- Detection of hepatitis C RNA and antibodies
- Proportion with hepatitis C given direct-acting antiviral treatment (DAAs)
- Cirrhosis measurement and monitoring
- Liver cancer screening
- Screening for chemsex use.
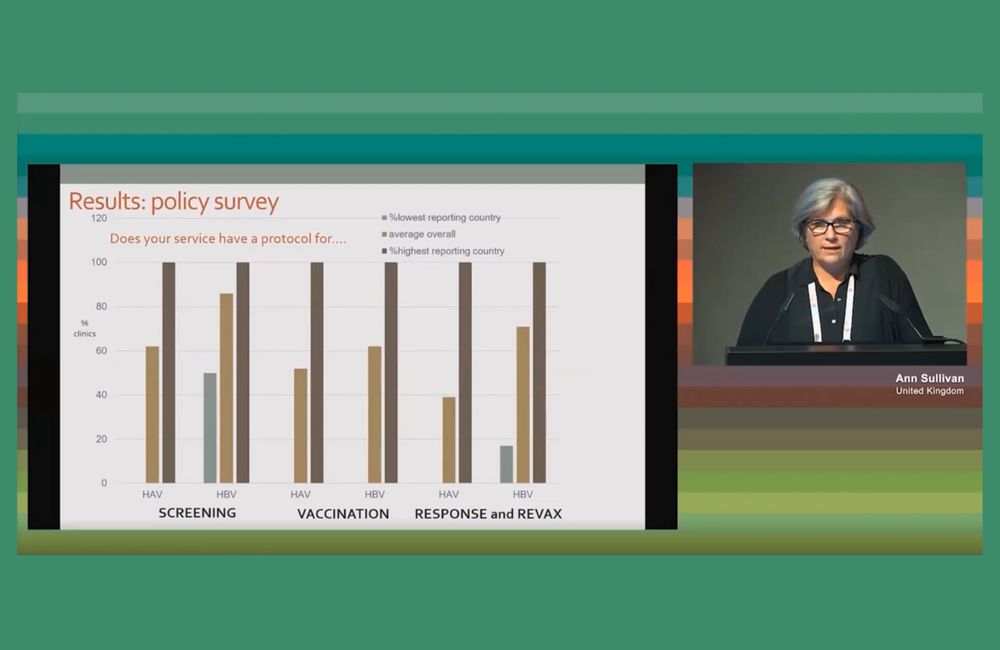
Regarding policies for hepatitis screening and vaccination, there was extremely wide variation between countries. In some countries, all services had protocols saying that all patients for HAV and HBV should be screened; at least one country did not have a policy of offering HAV screening at all. The lowest proportion of individual clinics in a country having a protocol for HBV screening was 50% and the lowest proportion having a protocol for liver cancer screening was 32%.
In more detailed case note reviews covering the care that was actually provided, a total of 610 patients’ case notes were reviewed: 329 new patients without hepatitis to measure screening and (where appropriate) vaccination rates; 135 patients co-infected with HBV, and 146 with HCV.
All patients had had at least one CD4 count recorded. Sullivan commented that it was unusual for any audit to find a clinical indicator with 100% performance, due to patients being lost to follow-up, information not being recorded on a patient’s notes, and so on, so this must be classed as an excellent result.
The proportion with a CD4 count within 30 days of HIV diagnosis was more variable, and in two clinics in different countries, only 50% had their CD4 count measured within this time. However, illustrative of the variability uncovered in the audit, another clinic in one of these two countries was one of the only clinics in the whole audit where 100% of their patients had a CD4 count within 30 days of diagnosis.
With hepatitis C screening, results were on the whole excellent. In two countries, all patients in two countries had been screened. In the lowest-performing country, only five patients out of 100 missed out on an HCV screen.
With hepatitis B screening, most patients had had the hepatitis B surface antigen test, which indicates current infection, acute or chronic. The lowest-performing country had screened 84% of patients, though two individual clinics in that country had screened all their patients. Most patients also received a hepatitis B core antigen test, which says whether you are or have been infected, but one country performed this test less often (28% of their patients).
Hepatitis A antibody screening, to check if people had had it and were immune or needed a vaccine, was less common. While all patients in one country received an HAV screen, only 7% in another country and 36% in another had received one. Only four clinics in the audit offered it to 100% of patients. However, Sullivan noted that HAV screening was one example of a standard that was affected by local practice: hepatitis A vaccination was not reimbursable under some countries’ healthcare systems, and in those cases there was little point in finding out if patients were immune to it or would benefit from vaccination.
"The purpose of the audit was to enable clinics to discover where their own practice was better or worse than in comparable clinics, not for EACS to measure their performance against external guidelines."
There was similar variability with hepatitis delta (HDV – the ‘satellite’ infection that can accompany HBV and make the development of cirrhosis and liver cancer more likely). While no patients in one country had been screened for HDV, and no patients in two individual clinics each in two other countries, one clinic each in those two countries performed HDV screening on all their patients who had HBV. Since HBV/HDV co-infection is the condition with the most serious outcomes for viral hepatitis, Sullivan described these results as “disappointing”.
Vaccination rates for HAV and HBV were more challenging to interpret as the immunity rate for the two viruses, indicating previous infection, varied hugely from clinic to clinic. In general, only a low proportion of patients had HAV vaccination which, as already noted, is not reimbursable in some countries. Rates of HBV vaccine reached 60% in one clinic but were generally in the region of 5 to 25%. However, in some countries, patients are usually referred to other services for HBV vaccination, making this data hard to collect. In four clinics, all patients selected for the case note review had immunity to HBV and must therefore have been infected or vaccinated in the past.
In contrast, there were encouraging rates of HCV treatment: 80% of co-infected HCV patients had received DAA treatment and it was planned for most of the others. Exceptions were one clinic in one country which had only treated half of its co-infected patients and had no plans for the others, and another clinic in another country which had treated 40% of co-infected patients with DAAs, they were planned for the others. What constituted ‘planned’ treatment, however, varied by definition between clinics.
Most clinics – 16 out of 23 – gave all their HBV co-infected patients ART that included tenofovir, which protects against chronic hepatitis B symptoms, but in one clinic no patient with HIV/HBV co-infection was on record as receiving a tenofovir-containing regimen, and in a couple of others only about half of them were.
The audit also covered asking patients about chemsex, but results varied considerably due to different proportions of gay and bisexual men in services. There is sometimes no standard practice of recording clinical discussions with patients – and if they are not recorded, they are regarded as not having happened by an audit.
Ann Sullivan concluded that a multicentre, multinational pan-European audit is feasible and acceptable to clinicians; it would allow national, regional and pan-European comparison of performance against EACS standards; and it would allow services to benchmark themselves to similar services in the same country – or in other countries. She emphasised ‘themselves’: the purpose of the audit was to enable clinics to discover where their own practice was better or worse than that in comparable clinics, not for EACS to measure their performance against external guidelines.
The challenges were that this audit, modest in size and time frame though it was, was undertaken entirely in clinicians’ spare time and was processed manually. Investment in an IT system would be necessary to enable a full-scale audit programme.
The conclusions of the pilot audit would be sent to the HIV heads of the relevant countries. If EACS finds the necessary resources, the audit will either be repeated on a larger scale, or different subjects might be audited such as late diagnosis or TB screening and treatment.
European AIDS Clinical Society. Standard of Care for HIV and Coinfections in Europe - Virtual, Tbilisi 2020. Click on this link for programme and all presentations.
The second part of the Tbilisi meeting was concerned with COVID-19 and its impact on HIV services; this subject was also covered in a workshop on 10 November and will be reported soon.
