Antiretroviral treatment containing the older formulation of tenofovir (tenofovir disoproxil fumarate; TDF) did not increase the risk of chronic kidney disease in previously untreated people with HIV at low risk for kidney disease, a large American cohort study reported last week at the 17th European AIDS Conference in Basel, Switzerland.
However, analysis of a large randomised trial carried out in South Africa showed that the newer formulation of tenofovir (tenofovir alafenamide; TAF) was associated with a significantly lower risk of reduced kidney function. The study also found that treatment containing TAF was associated with a lower risk of osteopenia (bone loss).
Chronic kidney disease in the OPERA cohort study
TDF, the older formulation of tenofovir, has been associated with an increased risk of developing chronic kidney disease, especially in people who receive tenofovir as part of a combination that includes a boosting agent (ritonavir or cobicistat). Tenofovir is known to have a harmful effect on the kidney tubules and boosting agents may raise levels of tenofovir in the kidneys, exacerbating the harmful effects of the drug.
TAF is a newer formulation which is less likely to cause kidney dysfunction. However, it is unclear if a switch to TAF is necessary in all patients. Given the increasing availability of low-cost generic versions of TDF, it is important to know whether some groups of people can continue taking TDF without increased risk of chronic kidney disease.
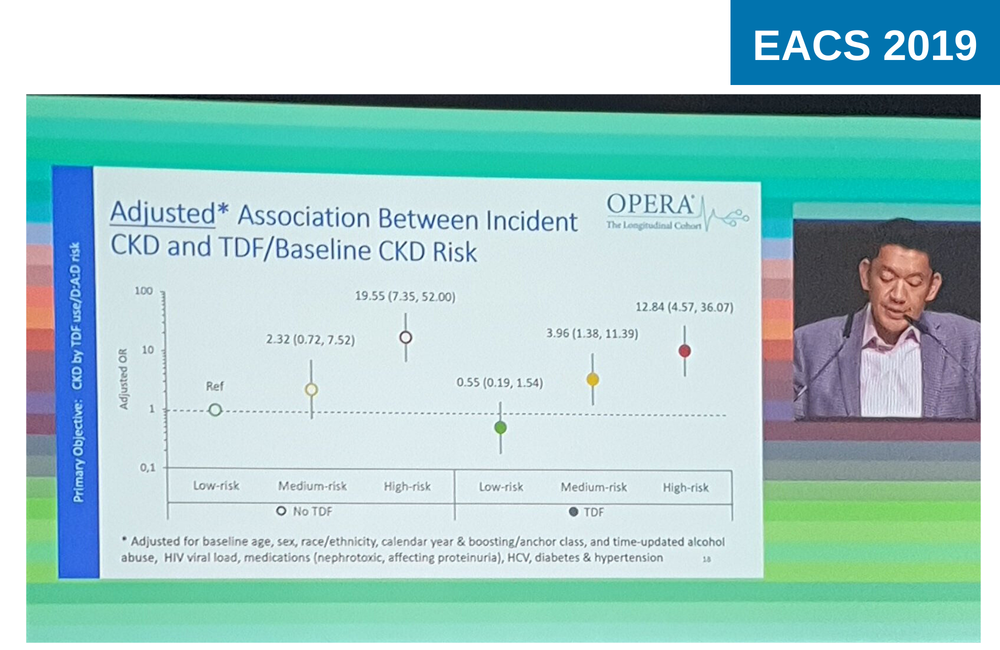
To investigate the risk of chronic kidney disease, US researchers looked at 9802 people who started antiretroviral treatment with normal kidney function (eGFR ≥60 ml/min/1.73m2 within 12 months prior to starting treatment). The cohort was stratified by prior risk of developing chronic kidney disease according to a scoring system developed by the D:A:D cohort study and clinical trial investigators, which draws on known, non-antiretroviral risk factors for chronic kidney disease.
Some antiretroviral drugs including dolutegravir, rilpivirine and cobicistat inhibit tubular creatinine secretion without harmful effects, leading to a lower eGFR result. In this study, the investigators corrected eGFR scores according to the reductions in eGFR observed in trials of third agents in the patient regimen.
In this cohort, 6222 people started treatment with a regimen containing TDF and were followed for median of 29 months, while 3580 started a non-TDF containing regimen and were followed for a median of 19 months.
There was very little difference in the proportion of people at medium or high risk of chronic kidney disease among those who included or avoided TDF in their first-line regimen (8% at high risk in both groups, 16% of the TDF group and 13% of the non-TDF group at medium risk), but those with medium and high risk were older, more likely to be women and initiated antiretroviral therapy earlier, regardless of TDF use.
People not taking TDF were more likely to be taking an integrase inhibitor; people taking TDF were more likely to be taking a non-nucleoside reverse transcriptase inhibitor.
In the low risk group, the incidence of chronic kidney disease (two eGFR measures < 60, at least 90 days apart) did not differ according to exposure to TDF (incidence rate 0.4 cases per 1000 person-years in both the TDF and non-TDF groups).
Chronic kidney disease developed most frequently in people at highest risk of chronic kidney disease at baseline (incidence rate 12.4 cases per 1000 person years of follow-up in those not exposed to TDF and 18.6 cases per 1000 person years).
People at high risk of kidney disease not taking TDF were almost 20 times more likely to develop chronic kidney disease during the follow-up period (adjusted odds ratio 19.8, 95% CI 7.35-52.00) compared to those at low risk. Among those taking TDF, those at highest risk were almost 13 times more likely to develop kidney disease compared to those at low risk (aOR 12.84, 95% CI 4.57-36.07)
There was no difference in the incidence of chronic kidney disease by TDF use or by use of a boosting agent.
Changes in markers of kidney function and bone loss in the ADVANCE trial
The ADVANCE study compared efavirenz with two dolutegravir-containing regimens, one paired with TDF, the other with TAF. The 96-week study monitored kidney function by measuring creatinine and tubular markers of kidney function (beta 2 microglobulin, retinol binding protein and the ratio of urine albumin to creatinine). The study monitored bone mineral density by DXA scan at baseline, week 48 and week 96 in all participants.
The incidence of treatment-emergent osteopenia in the hip was lowest in the TAF/emtricitabine+dolutegravir arm at week 96 (6.3%), compared to 11.1% in the TDF/emtricitabine+dolutegravir arm and 30% in the TDF/emtricitabine/efavirenz arm (p < 0.001). Treatment-emergent osteopenia in the spine was lowest in the TAF arm (18%) versus 23% and 30% in the TDF and efavirenz arms respectively, but this difference was not statistically significant.
There was no difference in the ten-year risk of hip fractures in participants aged 40 and over between the three study arms but participants in the efavirenz arm had a significantly higher ten-year risk of major fractures (p = 0.05). However, the absolute ten-year risk of a major fracture was 0.36% in the efavirenz arm.
In people with low bone mineral density, people with low body mass are at higher risk of fractures than people with higher body mass. "Any weight gain associated with antiretroviral treatment would ameliorate the increased fracture risk," Dr Michelle Moorhouse said.
There was no significant difference in grade 3 or 4 creatinine elevations or chronic kidney disease adverse events between study arms, but TAF recipients were significantly less likely to experience elevations above the upper limit of normal in tubular markers of kidney dysfunction.
Hsu R et al. Incidence of CKD with TDF and non-TDF containing antiretroviral regimens by baseline D:A:D CKD risk in people living with HIV (PLWH). 17th European AIDS Conference, Basel, abstract PS4/2, 2019.
Update: Following the conference presentation, this study was published in a peer-reviewed journal:
Hsu R et al. Risk of chronic kidney disease in people living with HIV by tenofovir disoproxil fumarate (TDF) use and baseline D:A:D chronic kidney disease risk score. HIV medicine, November 2020.
https://doi.org/10.1111/hiv.13019
Qavi A et al. The ADVANCE trial: the impact of DXA-assessed bone mineral density of TDF/FTC/EFV and TDF/FTC+DTG versus TAF/FTC+DTG. 17th European AIDS Conference, Basel, abstract PS4/3, 2019.

