The recent death of reality TV star Jade Goody has created a huge upsurge of concern about cervical cancer, and getting screened for it. HIV-positive women are especially likely to benefit from early and regular testing and treatment, reports Gus Cairns.
Cervical cancer is the second most common type of cancer in women worldwide, and the most common cause of cancer-related deaths in the developing world.1 If you develop invasive cervical cancer you have a less than 50% chance of survival.2
But Jade was unlucky to die from it. Cervical cancer is rare in women of her age and she may have had a less common variant of it. There are fewer than 30 deaths a year due to cervical cancer in women under 25 in the UK.
Over a lifetime, though, about one in 117 women will develop cervical cancer in the UK.3 Survival rates are continuing to improve: about two-thirds of women in England and Wales who develop cervical cancer will live for more than ten years after diagnosis.4
Most deaths from cervical cancer are preventable. The NHS calculates that if overall coverage of 80% were achieved with their cervical screening programme, there would be an eventual 95% reduction in cervical cancer deaths.5
One group of women who might particularly benefit from having more frequent cervical screening is women with HIV. HIV-positive women are twice as likely to be infected with HPV, the virus that causes cervical cancer, than HIV-negative women. They’re three to four times as likely to develop pre-cancerous cells if they are. And they are about twelve times as likely to develop invasive cervical cancer unless, in a simple procedure under local anaesthetic, these cells are removed.
What causes cervical cancer?
Cervical cancer is caused almost entirely by an infection – that of the human papilloma virus, HPV. HPV also causes a probable 90% of anal cancer cases and a minority of cases of cancer of the vagina, penis, and mouth. Only a small minority of people infected by HPV will develop cancer.
HPV is a whole family of viruses and there are about 100 different species, but just two types, HPV-16 and HPV-18, cause at least two-thirds of all cases of the most common type of cervical cancer. This is because they persist for longer and because they have acquired the ability to inactivate cancer-suppressing proteins normally produced by cells. For this reason the two highly effective vaccines developed in recent years, Cervarix and Gardasil, protect against these two types (Gardasil also protects against the two most common types that cause non-cancerous genital warts).
HPV is an extremely common infection. Three-quarters of HIV-negative women will catch at least one variety of HPV in their lifetime and half of them within two years of starting sex. However only 10-20% have a current infection and unless you have compromised immunity the body eventually gets rid of most HPV infections.
“It takes anything from a few weeks to 18 months for the body to produce antibodies (neutralising proteins) to HPV,” says Margaret Stanley, Professor of Epithelial Biology at Cambridge University. “Once this happens the immune response gets rid of the infection in 80 to 90% of cases. It’s the women who have persistent infection who are the ones at risk of developing cancer.”
The risks for HIV-positive women
Because a weak immune system controls HPV less well, women with HIV are at a disadvantage when it comes to rates of infection, the persistence of infection, and progression (the development or worsening of HPV-related conditions) and so need sooner and more frequent monitoring and earlier intervention.
Whereas only 15% of HIV-negative women may have current HPV infection, a large study in the USA found that nearly 60% of HIV-positive women were currently infected with at least one type of HPV.6
A study in pre-antiretroviral therapy (ART) days7 called the Women’s Interagency Health Study (WIHS) looked at a group of HIV-positive and negative women. It found that one in 25 HIV-negative women a year acquired new HPV infections but that the rate was 80% greater in women with HIV and three times greater in women with CD4 counts under 200 cells/mm3. Once they were infected, women with HIV with a high viral load were 50% more likely to develop SIL, or to develop high-grade SIL if they already had SIL. On the other hand, there was no statistical difference in the study between HIV-negative women and HIV-positive women with high CD4 counts and low viral loads.
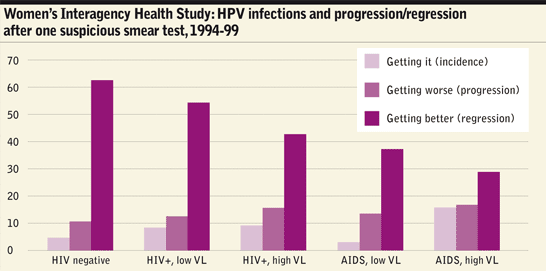
So does taking ART in itself reduce HPV infections and cervical cancer? Here the evidence is mixed: women on ART seem to clear more HPV infections but some surveys have shown no reduction in rates of SIL and CIN. This is probably because more women are living long enough now to develop persistent HPV infection; previously, many were dying of AIDS before they could develop it.
Screening and testing
Screening and treatment for pre-cancerous cells is a particular priority for women with HIV, since SIL can develop so much faster.
In the USA the Centers for Disease Control has recommended that HIV-positive women should be given a smear test as soon as they are diagnosed with HIV and then six months later, and every year after that if test results are normal.
Given however that a possibly much higher proportion of women with HIV who have high-risk HPV types will progress to SIL and CIN, changes such as including HPV DNA testing are being considered.8 This is a viral load-type test that can detect any current HPV infection, not just ones that produce symptoms, and can determine what type of HPV is present.
In the UK, the BHIVA sexual health guidelines (published 2008) recommend an initial smear test and colposcopy at HIV diagnosis followed by a smear test every year.9 They add that “CIN should be aggressively screened for and treated”. They do not recommend HPV DNA testing.
Dr Fiona Boag of the Kobler Clinic at the Chelsea and Westminster Hospital in London says that there is confusion in the minds of both patients and clinics about how often to screen HIV-positive women and who should do it.
“Women will be screened on HIV diagnosis but clinics are only now getting round to offering annual screening, and the uptake isn’t as good as it should be. We did a survey10 which showed that about two-thirds of our HIV-positive female patients were aware that they needed an annual screen, but that only half of them had actually had a screen in the last year.”
“Patients get confused because they also get letters from their GPs for three-yearly screening, as well as from us,” she adds.
In England the NHS took the controversial decision in 2005 only to offer screening to women over 25, but Boag confirms that HIV-positive women under 25 are also offered annual screening, “though some clinics still aren’t aware of the BHIVA guidelines”. So it may be necessary to be proactive to make sure you get your annual screening test.
The screening test for cervical abnormalities involves inserting a small spatula into the vagina and brushing a sample of cells from the cervix. This is often still called a ‘smear’ even though doctors now use a technique called liquid-based cytology (LBC) that involves putting the sample in a bottle rather than on to a microscope slide.
The test looks for abnormal cells that have been ‘excited’ by HPV. This is so-called SIL. If this is detected, a colposcopy will be ordered; this involves a doctor making a visual examination of the cervix (while the woman is in stirrups) with a binocular microscope. What they will be looking for is cervical intraepithelial neoplasia (CIN).
If it is left unchecked, then the CIN may develop firstly into a localised ‘pre-cancer’ called CIS (carcinoma in situ), which can still be operated on locally, and then into invasive cervical cancer needing radical treatment.
It’s important to emphasise that even if you develop CIN, malignant cervical cancer is not an inevitability. “If I knew why some women develop cancer and others don’t, I’d be on the plane to Stockholm to get my Nobel Prize,” says Margaret Stanley. Although about one in six women with HIV and HPV have either LSIL or HSIL (compared with one in 20 HIV-negative women), only about one in 400 HIV-positive women (0.24%) would develop invasive cervical cancer, which can take ten to 15 years to develop, if not treated.
Symptoms and treatment
CIN and cervical pre-cancer are more often than not asymptomatic, but if CIN is not detected they can produce symptoms. The most frequent one is bleeding between periods or after sex, or at any time if you are post-menopausal. Other symptoms include a smelly vaginal discharge, pain during sex and pain in the pelvic area in general.
Treatment involves a variety of methods according to the stage reached. If you have SIL and possibly CIN stage one, there may be no treatment advised other than follow-up tests, though you may be more likely to be offered treatment if you have HIV. Treatment for CIN may involve removal of the cells by non-surgical means such as cryotherapy (freezing with liquid nitrogen) or laser ablation (burning off the cells with a laser). Laser treatment is somewhat more effective and more usually offered. This treatment is usually done using local anaesthetic on an outpatient basis.
Simple surgical techniquesinclude loop excision involves the removal of a layer of precancerous cells with a ‘laser knife’ while cone biopsy involves the laser removal of a deeper layer of cells. These are minor day-surgery procedures. They are usually done using local anaesthetic though a general may be needed.
You may feel some discomfort during these procedures but they should not be painful after the initial local anaesthetic injection, though you may experience period-like pain and feel generally unwell for a few hours afterwards. There may also be some discharge or bleeding for a few days. Women are advised not to have sex, nor to use tampons, for four weeks after these procedures to allow the cervix to heal.
Studies have found that CIN is more likely to reappear in women with HIV than in HIV-negative women: one study11 found there was an 8.6% chance of high-grade CIN reappearing within a year. This was associated with low CD4 counts and not taking HIV treatment and also with incomplete removal of cells. The BHIVA cancer guidelines12 therefore recommend that doctors should err on the side of caution and use procedures that will ensure all cancerous cells are gone.
For persistent high-grade CIN and CIS, hysterectomy (removal of the womb) may be the treatment of choice. With more invasive cancers, this may have to be supplemented by radiotherapy, chemotherapy and possibly other surgery.
The vaccine
Finally, is it worthwhile getting vaccinated against HPV? After the success of the trials of the HPV vaccines, which prevented 95 to 100% of infection13 the UK, like many other countries, has recommended that either Gardasil or Cervarix be given to all adolescent girls; in the UK this means those aged 12 to 14.
It has been assumed that getting the vaccine if you are HIV positive is a waste of time, as so many women with HIV already have HPV, and are much more likely to be infected with multiple types than HIV-negative women.14 However even in HIV-positive women the body’s own antibody response may eventually get rid of those infections. Is it worth getting the vaccine in those circumstances?
Margaret Stanley thinks it is. “If I was positive I might well think about seeing which types of HPV I had and if I didn’t have types 16 or 18, getting the vaccine.” There is also evidence that the vaccines prevent infections by a quarter to a third of other high-risk HPV subtypes.15
At present you would have to get the HPV vaccine privately – where it costs in the region of £420 for the three-jab course. Nonetheless, some women are coming forward for it. Dr José González-García of the private sexual health and gay men’s clinic Freedom Health told HTU that there had been a 20% rise in women coming forward for HPV check-ups and cervical screening, both in his private and NHS work. The vaccine, however, will only protect the women who do not currently have HPV types 16 and 18.
Recommendations
The main recommendations for positive women are therefore:
- have a screening test on diagnosis and then at least once a year, and don’t wait for the clinic to send you reminders
- make sure you get the results (no news isn’t necessarily good news) and take up any follow-up screening or treatment to have any suspect cells removed promptly
- see if your clinic will provide an HPV DNA test to see what types of HPV you have. If you don’t have types 16 or 18, consider the vaccine.
If you do all these things, you suffer little risk of getting an almost entirely preventable disease and living to the ripe old age that Jade never had the chance to see.
1. De Vuyst H et al. HIV, human papillomavirus, and cervical neoplasia and cancer in the era of highly active antiretroviral therapy. Eur J Cancer Prev 17:545-554, 2008.
2. See www.cancerscreening.nhs.uk/cervical
3. See www.nhs.uk/Conditions/HPV-vaccination/Pages/Introduction.aspx
4. See www.cancerhelp.org.uk/help/default.asp?page=9260
5. See www.cancerscreening.nhs.uk/cervical
6.Palefsky JM et al. Cervicovaginal human papillomavirus infection in HIV positive and high risk HIV negative women. J Natl Cancer Inst 91:226-236, 1999.
7. Massad LS et al. Evolution of cervical abnormalities among women with HIV-1: evidence from surveillance cytology in the Women’s Interagency Health Study. J Acquir Immune Defic Syndr 25(5):432-442, 2001.
8. Sawaya GF et al. Risk of cervical cancer associated with extending the interval between cervical cancer screenings. NEJM 349:1501-1509, 2003.
9. Fakoya A et al. British HIV Association, BASHH and FSRH guidelines for the management of the sexual and reproductive health of people living with HIV infection 2008. HIV Medicine 9:681-720, 2008.
10. Cooney G et al. Understanding the sexual and reproductive health needs of women living with HIV. Fifteenth BHIVA Conference, Liverpool, poster presentation P19, 2009.
11. Heard I et al. High rate of recurrence of cervical intraepithelial neoplasia after surgery in HIV-positive women. J Acquir Immune Defic Syndr 39:412-418, 2005.
12. See www.bhiva.org/files/file1030995.pdf
13. The FUTURE II study group. Effect of prophylactic human papillomavirus L1 Virus-like-particle vaccine on risk of cervical intraepithelial neoplasia grade 2, grade 3 and adenocarcinoma in situ: a combined analysis of four randomised clinical trials. Lancet 369:1861-8, 2007.
14. Clifford GM et al. Human papilloma virus types among women infected with HIV: a meta-analysis. AIDS 20:2337-2344, 2006.
15. Brown DR et al.The impact of quadrivalent human papillomavirus (HPV; types 6, 11, 16, and 18) L1 virus-like particle vaccine on infection and disease due to oncogenic nonvaccine HPV types in generally HPV-naive women aged 16-26 years. J Infect Dis 199: online publication, April 2009.
