Metabolic syndrome – including obesity – developed significantly more frequently in people who received dolutegravir, tenofovir alafenamide (TAF) and emtricitabine compared to other regimens in the South African ADVANCE study, Dr Michelle Moorhouse reported on Thursday at the 17th European AIDS Conference in Basel, Switzerland.
Furthermore, updated follow-up from the study showed that close to 60% of women in the TAF arm experienced a weight gain of at least 10% during the 96-week study. By the end of the follow-up period, approximately 75% were overweight or obese.
Forty-eight week efficacy results of the ADVANCE study were presented at the 10th IAS Conference on HIV Science in Mexico City in July, with 96-week data showing changes in body weight and body weight distribution.
This week, study investigators reported 96-week body composition results on a larger proportion of participants enrolled in the trial and also reported on the incidence of metabolic syndrome during the study.
The ADVANCE study is a large, randomised comparison of:
- dolutegravir/tenofovir disoproxil fumarate/emtricitabine (DTG/TDF/FTC)
- dolutegravir/tenofovir alafenamide/emtricitabine (DTG/TAF/FTC)
- efavirenz/tenofovir disoproxil fumarate/emtricitabine (EFV/TDF/FTC).
The study was designed to evaluate the efficacy and safety of dolutegravir in a South African population as the country prepared to introduce the drug, and to evaluate whether a nucleoside backbone containing the newer formulation of tenofovir (TAF) reduced the risk of kidney and bone toxicities in the South African setting.
Forty-eight week efficacy results showed that both dolutegravir-based regimens were non-inferior to the efavirenz-based regimen. However, the study also found a higher incidence of weight gain – as measured by changes in body mass index (BMI), absolute weight and emergence of obesity – in people who received dolutegravir, especially in those randomised to receive DTG/TAF/FTC.
This week, Dr Michelle Moorhouse of Ezintsha (Wits Reproductive Health and HIV Institute), Johannesburg, presented further information on changes in body composition in 531 participants who have completed 96 weeks of follow-up (1053 participants were randomised, 169 have discontinued, 77 became pregnant during the study and 276 have missing data).
Approximately 60% of participants were women, 99% were black and the mean CD4 cell count ranged from 323 to 349 across the three study arms. At randomisation, 35 to 37% according to study arm were already either overweight (BMI 25 to 30) or obese (BMI above 30).
Weight gain
As previously reported, the most pronounced gains in weight after 96 weeks after randomisation occurred in those receiving DTG/TAF/FTC (+6kg in men, +9kg in women). Body composition was assessed by DXA scan at weeks 48 and 96.
In men, weight gain at week 96 consisted entirely of fat in the efavirenz recipients (+1.2kg), evenly distributed between limb and trunk fat. In the DTG/TDF/FTC recipients, weight gain (+3.5kg) was 74% fat, again evenly distributed between limb and trunk fat. In the DTG/TAF/FTC recipients, a higher proportion of the total weight gained (+5.9kg) came in the form of lean mass (59% of total weight gain was fat). However, the proportion of weight gained in the form of fat increased from 53% to 59% in TAF recipients between weeks 48 and 96.
In women, weight gain consisted predominantly of fat in the efavirenz recipients (+3.4kg, 91% fat). In the DTG/TDF/FTC recipients, weight gain (+5.3kg) was 74% fat, again evenly distributed between limb and trunk fat. In the DTG/TAF/FTC recipients, weight gain (+8.3kg) was predominantly fat (74%), and almost all lean mass was gained in the limbs.
Treatment-emergent obesity and weight gain of 10% or more at week 96 were each more common in dolutegravir recipients and most frequently observed in DTG/TAF/FTC recipients.
Weight gain of 10% or more occurred in 51% of women and 42% of men receiving DTG/TAF/FTC, compared to 32% of women and 27% of men receiving DTG/TDF/FTC. In comparison, 23% of women and 18% of men in the efavirenz arm gained at least 10% of their baseline body weight.
“Most participants refused point blank to switch from dolutegravir – they don’t want to go back to efavirenz.”
Increases in weight gain in women were progressive and did not appear to plateau prior to 96 weeks, whereas in men weight gain was most pronounced up to week 48.
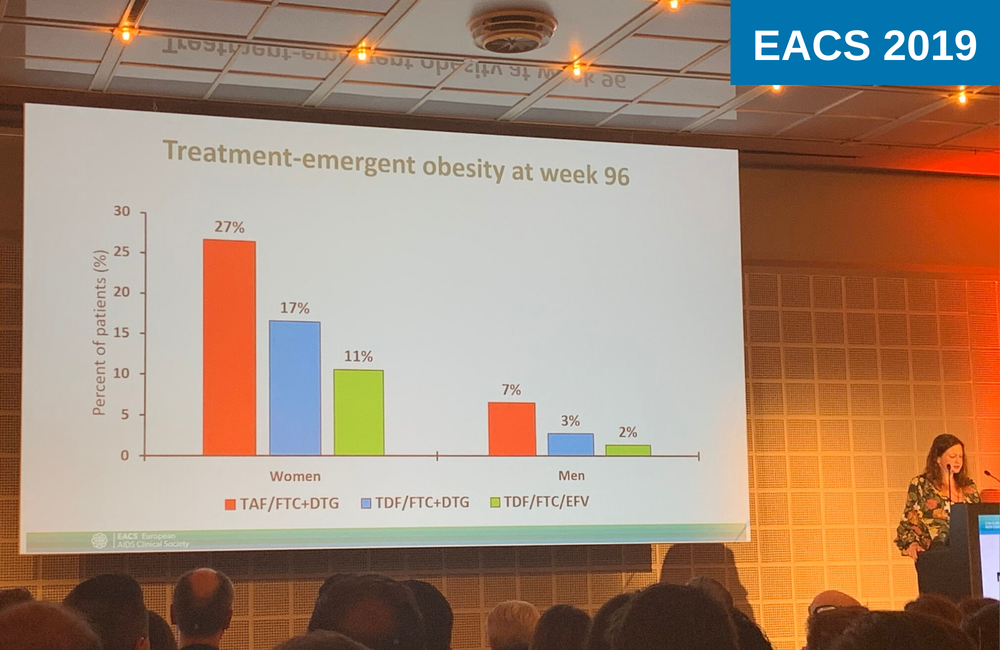
Treatment-emergent obesity – weight gain that placed a participant in the obese category (BMI >30) – was more common in women, but women had a higher mean BMI at baseline. Twenty-seven per cent of women and 7% of men became obese while taking the TAF-containing regimen, compared to 17% of women and 3% of men taking DTG/TDF/FTC and 11% of women and 2% of men taking the efavirenz-based regimen.
Multivariable analysis that controlled for sociodemographic factors, baseline HIV factors, metabolic disease history and concomitant medications found that treatment-emergent obesity was associated with: DTG/TAF/FTC, baseline CD4 count, baseline viral load, and baseline BMI. Weight gain of greater than 10% was associated with: DTG/TAF/FTC, baseline CD4 count, baseline viral load, female sex, age, and baseline weight.
Treatment tolerability and weight gain
The investigators also analysed whether gastrointestinal tolerability of treatment had any impact on weight gain. Some researchers have suggested that the reason for greater weight gain on newer regimens, especially those containing an integrase inhibitor and/or TAF, is because they have fewer gastrointestinal side effects, leading to better food absorption and appetite. The ADVANCE study investigators found no difference in weight gain in the dolutegravir arms when they excluded 77 people from the analysis who reported gastrointestinal side effects during the study, although weight gain was greater in the efavirenz group (+0.6kg).
Metabolic syndrome
The investigators also analysed the emergence of metabolic syndrome during treatment. Metabolic syndrome was defined as obesity plus any two of: elevated triglycerides (or treatment for); reduced HDL cholesterol (or treatment for); raised blood pressure (or treatment for); elevated glucose (or treatment for type 2 diabetes).
Treatment-emergent metabolic syndrome was observed in 9% of DTG/TAF/FTC recipients at week 96, compared to 5% of DTG/TDF/FTC recipients and 3% of EFV/TDF/FTC recipients (p=0.025 for difference TAF vs EFV arms).
Asked how trial participants were being managed in the light of the findings, Michelle Moorhouse said that all study participants had been informed of the weight gain results and asked to give renewed consent to remain in the study. For the time being investigators are “watchfully monitoring”, she said, but added that “most participants refused point blank to switch from dolutegravir – they don’t want to go back to efavirenz” owing to its central nervous system side effects.
McCann K et al. The ADVANCE clinical trial: changes from baseline to week 96 in DXA-assessed body composition in TAF/FTC +DTG compared to TDF/FTC+DTG, and TDF/FTC/EFV. 17th European AIDS Conference, Basel, abstract PS3/3, 2019.
Update: Following the conference presentation, this study was published in a peer-reviewed journal:
Venter W et al. Dolutegravir with emtricitabine and tenofovir alafenamide or tenofovir disoproxil fumarate versus efavirenz, emtricitabine, and tenofovir disoproxil fumarate for initial treatment of HIV-1 infection (ADVANCE): week 96 results from a randomised, phase 3, non-inferiority trial. The Lancet HIV, 7: e666-e676, October 2020.
