Around four in ten women and one in ten men taking injectable cabotegravir as pre-exposure prophylaxis (PrEP) still have evidence of the drug in their body around 18 months after their last injection, Raphael Landovitz of the University of California told the HIV Research for Prevention conference (HIVR4P 2018) in Madrid yesterday. While providing evidence of the durability and potency of injectable cabotegravir, the findings raise concerns about the potential development of drug resistance.
If people stop receiving PrEP injections, they will be vulnerable to HIV unless they start or continue another method of HIV prevention, such as oral PrEP. In addition, the ‘long tail’ in some people means that there is a lengthy period during which, if they caught HIV, they could develop drug resistance. Drug resistance only arises in situations like this when there is some drug in the body but not enough to fully suppress an infection.
The problem was first identified two years ago in the ECLAIR study, whose participants were all American men. In order to better understand the issue, follow-up in another study, HPTN 077, was extended from 48 to 76 weeks (approximately 18 months) after participants’ last injection. This phase II study was designed to examine the safety, tolerability and pharmacodynamics of injectable cabotegravir, but not its efficacy.
This study recruited 199 women and men in the United States, South Africa, Malawi and Brazil who did not have HIV and were at low risk of acquiring it.
Participants were randomly assigned in a 3-to-1 ratio to receive cabotegravir or placebo injections. They first received four weeks of cabotegravir pills to ensure safety, as the injectable drug cannot be removed if side-effects occur. One cohort received 800mg doses of injectable cabotegravir every 12 weeks. A second cohort received 600mg doses every eight weeks.
The study showed that the eight weekly dosing was likely to be more effective and it has been taken forward for further development. However, in terms of pharmacokinetics or safety, there were no significant differences between the two dosing schedules, so the cohorts were combined for the analysis presented in Madrid this week. Measurements of plasma drug levels were taken every 12 weeks after the final injection.
Analysis focused on 133 participants who received at least one injection of cabotegravir, including 102 who agreed to continue with follow-up visits through to 76 weeks.
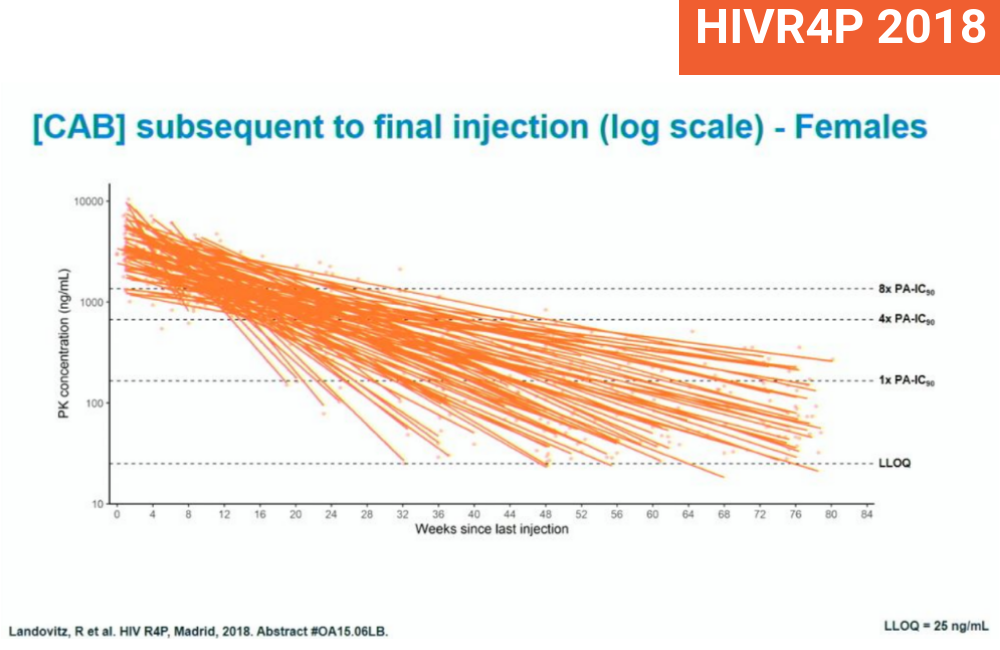
The half life is the time it takes for the concentration of drug in the body to reduce by one half. In women, the mean was 64 days, but was highly variable between participants, ranging from 19 to 217 days. In men it was 42 days, ranging from 13 to 134 days.
After adjusting for other factors, the half life in women was 45% longer than that in men. It was also longer for heavier people (2% increase for each body mass index [BMI] unit) – the amount of fat in the buttocks, where the injections are given, might be part of the explanation. Nonetheless, these factors only explained 17% of the variability in half lives.
The researchers also looked at the median time for drug levels to fall below the lower limit of quantification (LLOQ), in other words to no longer be detectable. In women, it was 66 weeks, ranging from 18 to 182 weeks. The latter figure indicates that some women continued to have detectable drug up to three and a half years after their last injection. In men, the median was 43 weeks, ranging from 20 to 134 weeks.
At 24 weeks after their last injection, 55% of women still had drug levels which – based on animal studies – are likely to protect against 98% of vaginal exposures (greater than 4 x IC90). Forty per cent still had detectable drug, but at a level that is likely to be sub-optimal, only protecting around 80% of vaginal exposures. Five per cent had no detectable drug (below the LLOQ).
At 76 weeks, no female participants had a high level of protection, 42% had sub-optimal levels and 58% had no detectable drug.
Turning now to men, at 24 weeks, 24% had a level thought to protect against 100% of rectal exposures (greater than 4 x IC90) and 27% had a level that would protect against 97% of rectal exposures (between 1 and 4 x IC90). A further 27% had sub-optimal levels, likely to only provide 74% protection. Twenty-two per cent had no detectable drug.
At 76 weeks, no male participants had a high level of protection, 13% had sub-optimal levels and 87% had no detectable drug.
Adverse events during the tail phase were comparable to those earlier in the study. There were no additional HIV seroconversions.Two participants became pregnant around 32 weeks and 108 weeks after their last injections, giving birth to healthy infants.
Landovitz did not speculate on what the data means for the future of cabotegravir as injectable PrEP. This is being more fully evaluated in two large phase III studies, HPTN 083 and HPTN 084, which have around 7000 participants between them and are expected to report results in 2020-21. In particular the studies will clarify whether the development of drug resistance is indeed a problem when HIV infections occur in people who have stopped receiving injections.
HPTN 083 and HPTN 084 participants are being asked to take oral PrEP for 48 weeks after their last injection to ‘cover’ the long tail, but will this be long enough? And will this be a feasible approach in routine clinical care?
Landovitz RJ et al. Tail-phase Safety, Tolerability and Pharmacokinetics of Long-acting Injectable Cabotegravir in HIV-uninfected Individuals: HPTN 077 Final Results. HIV Research for Prevention conference (HIVR4P 2018), Madrid, October 2018, abstract OA15.06LB
View the abstract on the conference website.
View the slides on the conference website (with audio).
Update: Following the conference presentation, the study was published in a peer-reviewed journal:
Landovitz RJ et al. Tail-phase safety, tolerability, and pharmacokinetics of long-acting injectable cabotegravir in HIV-uninfected adults: a secondary analysis of the HPTN 077 trial. The Lancet HIV, online ahead of print, June 2020.
https://doi.org/10.1016/S2352-3018(20)30106-5
