A combination of three direct-acting antivirals developed by Bristol-Myers Squibb cured chronic hepatitis C infection in over 90% of previously untreated patients in a mid-stage study, Gregory Everson of the University of Colorado reported at 'The Liver Meeting 2013', the 64th annual meeting of the American Association for the Study of Liver Diseases (AASLD) in Washington, DC, on Monday.
The combination proved equally effective in people with genotype 1a and 1b hepatitis C.
Bristol-Myers Squibb is one of several pharmaceutical companies working to develop a highly effective combination of oral direct-acting antivirals that can be used without interferon to cure hepatitis C infection with 12 to 24 weeks of treatment. Current hepatitis C treatment consists of pegylated interferon and ribavirin combined with a protease inhibitor, and lasts 24 to 48 weeks.
Interferon-free combinations contain drugs which attack different steps in the hepatitis C viral life-cycle, so that viral replication can be interrupted and quickly reduced, allowing rapid elimination of hepatitis C from the liver and the blood.
AbbVie, Boehringer Ingelheim, Gilead Sciences and Merck have also presented data on the progress of their own interferon-free combinations at this year’s AASLD conference.
The first interferon-free combination is likely to receive marketing approval for treatment of genotype 2 and 3 hepatitis C in December 2013 (sofosbuvir and ribavirin, manufactured by Gilead Sciences). AbbVie is likely to file for approval of its own interferon-free combination for treatment of genotype 1 infection in the United States and European Union in the second quarter of 2014.
Bristol-Myers Squibb is developing a fixed-dose combination containing drugs from three classes. Daclatasvir, an NS5A inhibitor active against all genotypes, is being combined with asunaprevir, a protease inhibitor active against genotypes 1, 4, 5 and 6, and BMS-791325, a non-nucleoside polymerase inhibitor active against genotypes 1, 3, 4, 5 and 6. The fixed-dose combination is designed to be dosed twice a day.
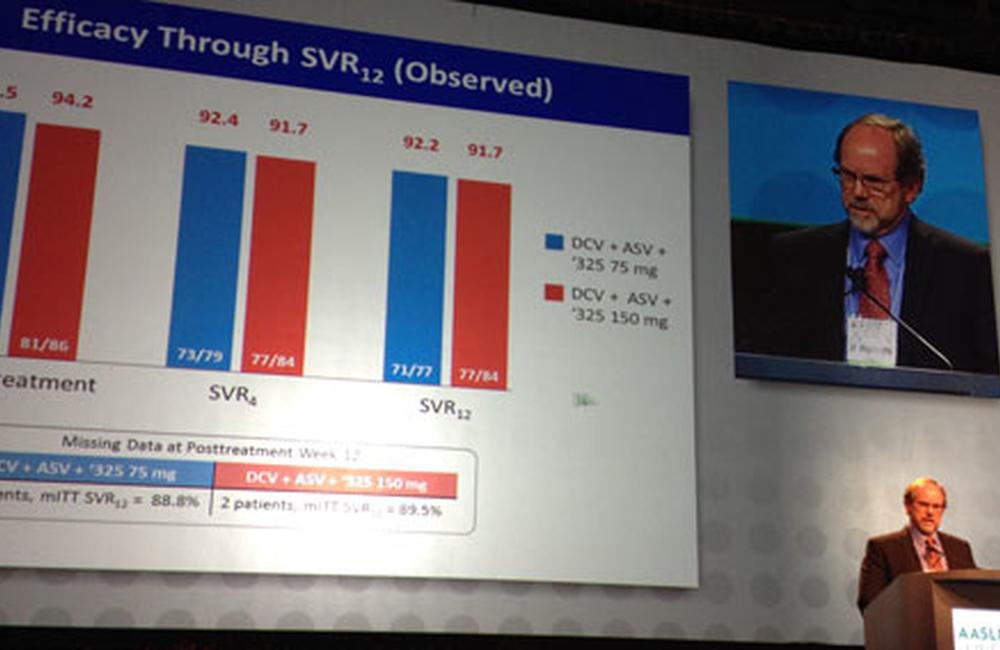
Gregory Everson presented interim results of a phase IIb dose-comparison study designed to compare two doses of BMS-791325 (75mg and 150mg) for the purpose of selecting a dose for phase III studies.
The study recruited previously untreated patients with genotype 1 infection. Nine per cent of patients had cirrhosis, and patients with cirrhosis were evenly distributed between the two study arms. Patients were also stratified by genotype 1a and 1b. Eighty patients were recruited to the 75mg arm and 86 to the 150mg arm.
Eight-two per cent of patients had genotype 1a hepatitis C infection and 38% had advanced liver disease (stage F3 or F4 fibrosis as measured by Fibroscan).
Twelve weeks after the completion of treatment 71 of 77 (92.2%) participants in the 75mg arm and 77 of 84 (91.7%) participants in the 150mg arm had a sustained virologic response (SVR12). Three people were lost to follow-up after completion of treatment. Six cases of virological failure occurred in the 75mg arm (2 viral breakthroughs and 4 post-treatment relapses) and five in the 150mg arm (3 viral breakthroughs and 2 viral relapses). All viral relapses occurred within four weeks of completing treatment.
The two doses showed equivalent efficacy across all sub-groups (sub-groups, genotype 1a and 1b, IL28B CC and non-CC genotype) with the exception of people with cirrhosis, where the 75mg dose was associated with a higher rate of SVR (100 vs 71%) due to one treatment discontinuation and one viral breakthrough.
The combination was well tolerated. One person in each arm discontinued treatment due to an adverse event (one cancer and one episode of severe throat tightness). One person experienced a grade 3 liver enzyme elevation (AST) which normalised during treatment.
The most frequent side-effects were headache (24.7%), diarrhoea (15.1%), fatigue (11.4%) and nausea (10.2%), none of them severe.
The 75mg dose of BMS-791352 will now be used as part of the three-drug combination in phase III studies.
Everson G et al. Phase 2b study of the interferon-free and ribavirin-free combination of daclatasvir, asunaprevir, and BMS-791325 for 12 weeks in treatment-naive patients with chronic HCV genotype 1 infection. 64th Annual Meeting of the American Association for the Study of Liver Diseases, Washington DC, abstract LB-1, 2013.
