A ‘functional cure’ of HIV has been achieved in one monkey given gene therapy that induced it to make two broadly neutralising antibodies against the virus.
Researcher José Martinez-Navio of the University of Miami told the HIV Research for Prevention conference (HIVR4P 2018) that the monkey received no antiretroviral therapy (ART) at any time other than two therapeutic vaccinations containing the genes that induce antibodies, and has remained with an undetectable viral load for nearly three years so far.
The experiment with a therapeutic vaccine, he said, was inspired by a previous experiment in which two monkeys were given a preventative gene-therapy vaccine that induced them to express an antibody called 5L7 against the monkey HIV equivalent, SIV. One of the two monkeys remained uninfected despite six intravenous challenges with highly infectious virus over a period of over 3.5 years. His team therefore decided if a similar vaccine could be used to cure an existing infection.
The 'Miami monkey' cannot be regarded as totally cured of its infection. The researchers were able to find viral RNA and DNA at low levels in the monkey’s body, at a level of three to 20 copies of viral RNA in a million T-cells and roughly the same level of integrated viral DNA. And in one of the most stringent tests of HIV infection, replicating HIV was recovered from cells taken from the monkey and cultured in a lab dish with an immune suppressant that destroyed all the CD8 cells essential for a functioning immune system. This was equivalent to reversing the therapeutic effects of the vaccine.
However, attempts to infect two other monkeys with transfusions taken from the first one resulted in no infection and the monkey must be regarded as essentially lacking active HIV.
The two antibodies used were the same two broadly neutralising antibodies (bNAbs), 10-1074 and 3BNC117, that were recently used in an experiment in humans in which seven out of eleven people without apparent pre-existing resistance to the antibodies maintained an undetectable viral load off ART for an average of five months.
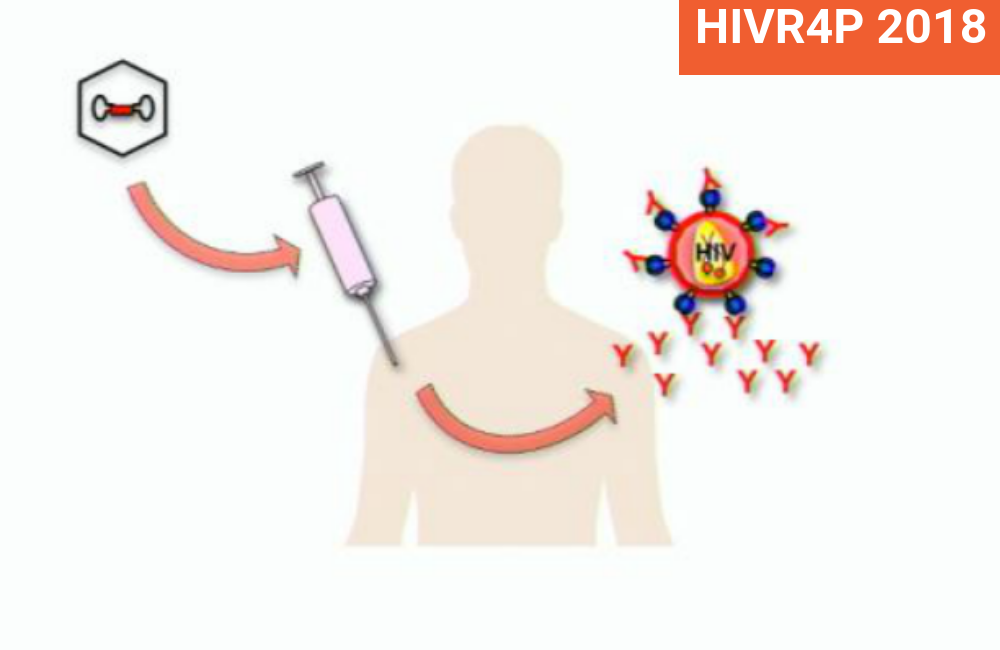
The difference with this experiment is that the antibodies were not passively infused directly but were given as a gene therapy. This consisted of two modified genes that were introduced into the monkey’s own immune system by means of a vector, the shell of an inert virus called AAV (adeno-associated virus) which does not cause disease but is able to insert the genes inside the host genome.
This induced the monkey’s immune system to express the bNAbs which, as the researchers say, “can result in continuous durable expression of the transgene product for years”. This is a true therapeutic vaccine, unlike the bNAbs in the human experiment that were simply infused and eventually eliminated from the body.
The AAV vaccine was designed to elicit three different bNAbs – the other one was called 10E8. The vaccine was given to four monkeys. However, in what is an ongoing problem for antibody-stimulating vaccines, the monkeys produced counter-antibodies that prevented the expression of 10E8 in all four cases, 3BNC117 in three cases, and 10-1074 in one case.
The monkeys were infected with a potent ‘humanised monkey HIV’ called SHIV-AD8. They received no treatment for 86 weeks (a year and seven months) before being given the AAV-vector vaccine.
The monkey that developed counter-antibodies to all three bNAbs and was therefore on a ‘functional placebo’ unfortunately died of its SHIV infection at week 100. Of the two only left with 10-1074, one had an essentially unchanged viral load; interestingly, though, the other monkey that only had 10-1074 developed a viral load that has remained undetectable (below 15 copies/ml) with occasional blips to no more than 150 copies/ml.
The fourth monkey, numbered rh2438, developed a viral load that was consistently undetectable and still is, at 242 weeks after SHIV infection and 158 weeks (just over three years) after AAV vaccination; it also maintains consistently high plasma levels of 10-1074 and 3BNC117.
As mentioned above, the monkey still appears to have HIV in some of its cells but the persistent levels of antibody and the presence of a functional anti-SHIV CD8 response indicate prolonged recession of HIV infection or even a permanent functional cure.
In a second experiment, the researchers repeated the vaccine design in eight monkeys, using an AAV vaccine expressing four bNAbs. Again two or three of these bNAbs were neutralised by counter-antibodies but three of the eight monkeys, that maintained reasonable levels of an antibody called PGT128, have also maintained undetectable viral loads interrupted by a few blips for a year after AAV inoculation.
These experiences are very much proofs of concept and researchers will have to find a way of preventing the immune system from making counter-antibodies to the bNAbs. But this is one of the first demonstrations that a vaccine designed to elicit specific, monoclonal broadly neutralising antibodies can produce a functional cure.
Martinez-Navio JM et al. Long-term delivery of anti-HIV monoclonal antibodies. HIV Research for Prevention conference (HIVR4P 2018), Madrid, 2018, abstract OA13.05.